QUESTION IMAGE
Question
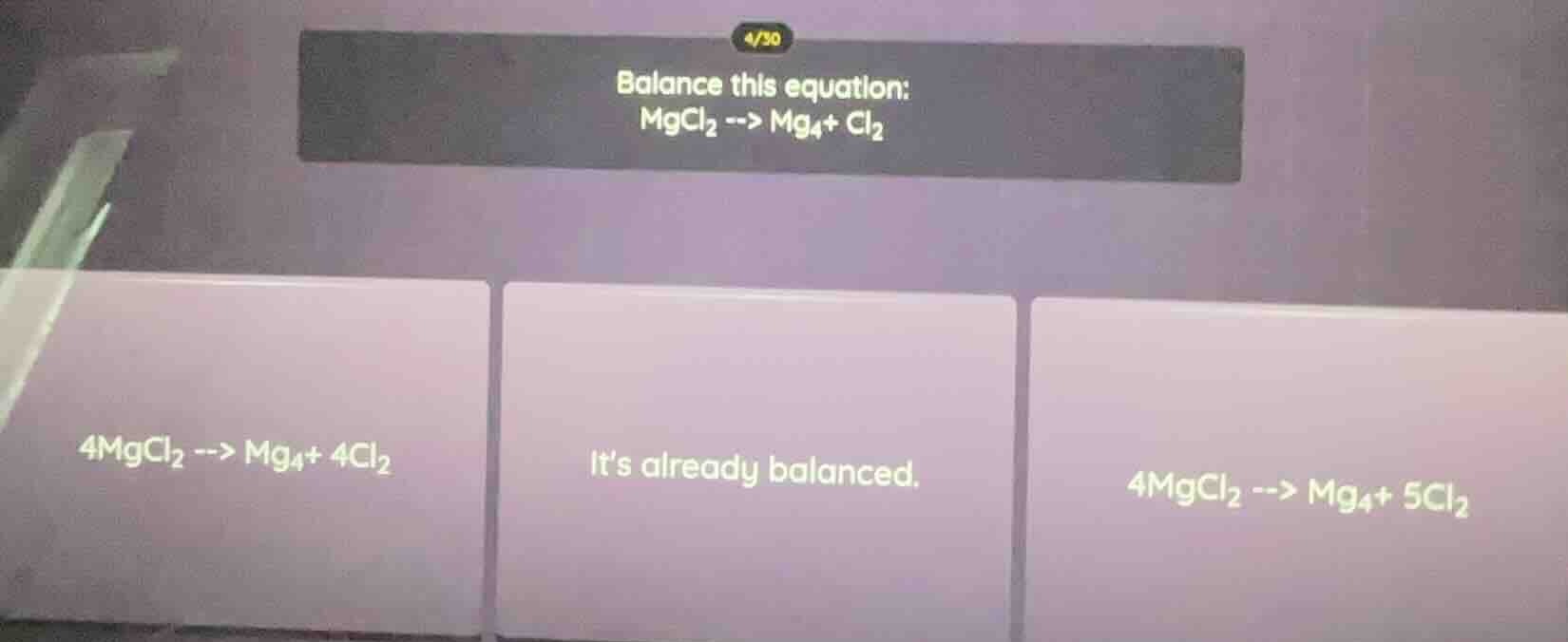
balance this equation: mgcl₂ --> mg₄+ cl₂ 4mgcl₂ --> mg₄+ 4cl₂ it’s already balanced. 4mgcl₂ --> mg₄+ 5cl₂
Step1: Count Mg on both sides
Reactants: 1 Mg in $\text{MgCl}_2$; Products: 4 Mg in $\text{Mg}_4$
Step2: Balance Mg atoms
Multiply $\text{MgCl}_2$ by 4: $4\text{MgCl}_2
ightarrow \text{Mg}_4 + \text{Cl}_2$
Step3: Count Cl on both sides
Reactants: $4 \times 2 = 8$ Cl; Products: 2 Cl in $\text{Cl}_2$
Step4: Balance Cl atoms
Multiply $\text{Cl}_2$ by 4: $4\text{MgCl}_2
ightarrow \text{Mg}_4 + 4\text{Cl}_2$
Step5: Verify balanced equation
Mg: 4 on both sides; Cl: 8 on both sides
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. $4\text{MgCl}_2
ightarrow \text{Mg}_4+ 4\text{Cl}_2$