QUESTION IMAGE
Question
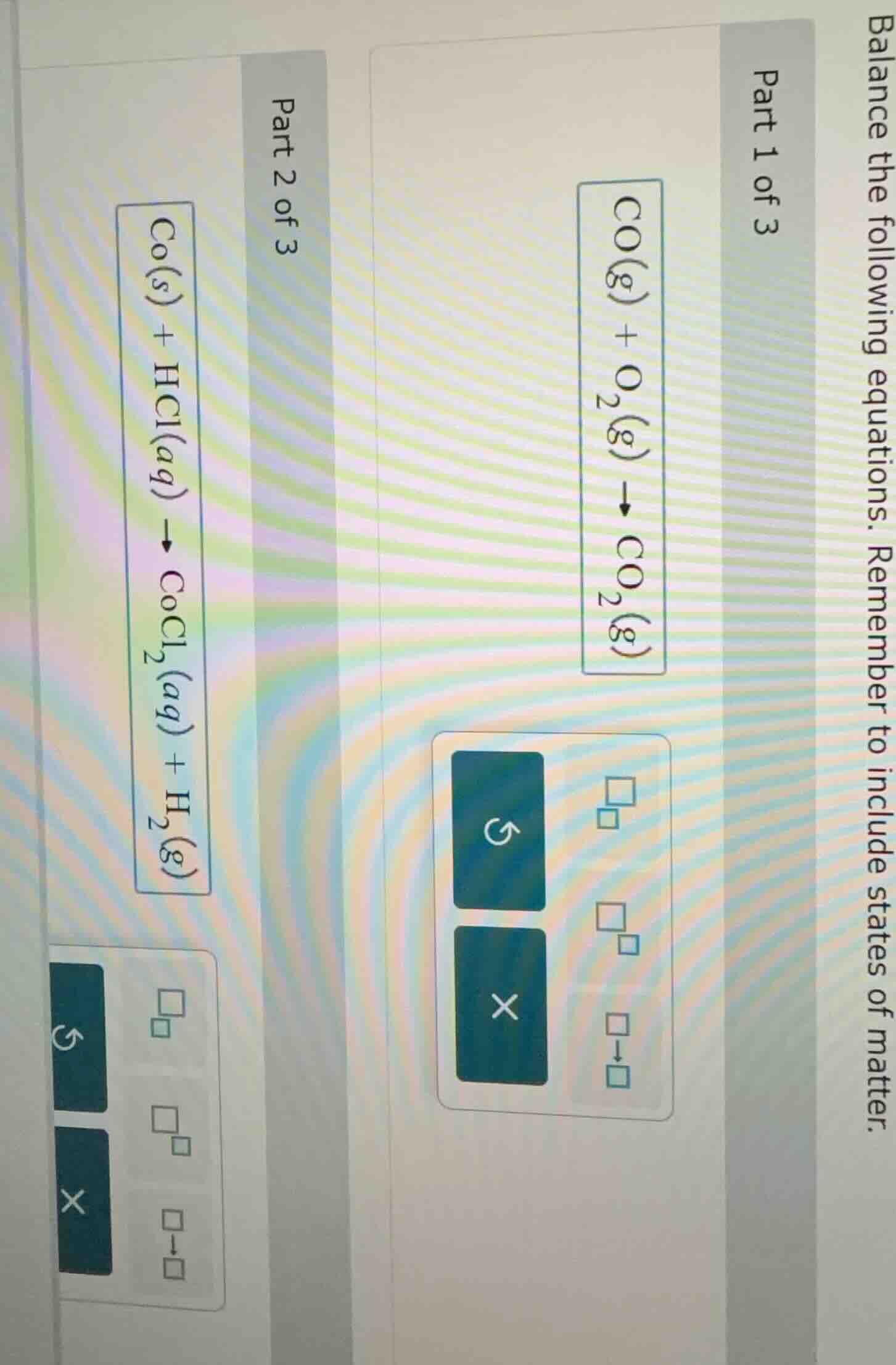
balance the following equations. remember to include states of matter.
part 1 of 3
$\ce{co(g) + o2(g) -> co2(g)}$
part 2 of 3
$\ce{co(s) + hcl(aq) -> cocl2(aq) + h2(g)}$
Part 1 of 3
Step1: Analyze carbon and oxygen atoms
On the left side, we have 1 C in \( \text{CO}(g) \) and 2 O in \( \text{O}_2(g) \). On the right side, we have 1 C and 2 O in \( \text{CO}_2(g) \). Let's start with balancing carbon first (it's already 1 on each side). Now for oxygen: left side has \( 1 + 2 = 3 \) O (wait, no, \( \text{CO} \) has 1 O, \( \text{O}_2 \) has 2 O, so total O on left is \( 1 + 2x \) where \( x \) is the coefficient of \( \text{O}_2 \), and on right, \( \text{CO}_2 \) has 2 O per molecule, with coefficient \( y \) for \( \text{CO}_2 \) and coefficient \( z \) for \( \text{CO} \). Wait, better approach: let's let the coefficient of \( \text{CO} \) be \( a \), \( \text{O}_2 \) be \( b \), and \( \text{CO}_2 \) be \( c \). So \( a\text{CO}(g) + b\text{O}_2(g)
ightarrow c\text{CO}_2(g) \). For C: \( a = c \). For O: \( a + 2b = 2c \). Since \( a = c \), substitute \( c \) with \( a \): \( a + 2b = 2a \) → \( 2b = a \). Let's choose \( a = 2 \), then \( b = 1 \), and \( c = 2 \).
Step2: Write the balanced equation
Substituting the coefficients: \( 2\text{CO}(g) + \text{O}_2(g)
ightarrow 2\text{CO}_2(g) \)
Step1: Analyze each element
For Co: left side has 1 Co in \( \text{Co}(s) \), right side has 1 Co in \( \text{CoCl}_2(aq) \). For Cl: left side has 1 Cl in \( \text{HCl}(aq) \), right side has 2 Cl in \( \text{CoCl}_2(aq) \). For H: left side has 1 H in \( \text{HCl}(aq) \), right side has 2 H in \( \text{H}_2(g) \).
Step2: Balance Cl first
To balance Cl, we need 2 HCl, so coefficient of \( \text{HCl} \) is 2. Now, H on left: 2 (from 2 HCl), so on right, \( \text{H}_2 \) needs coefficient 1 (since 2 H per \( \text{H}_2 \)). Co is already 1 on each side. Now check: Co: 1 = 1, Cl: 2 = 2, H: 2 = 2.
Step3: Write the balanced equation
\( \text{Co}(s) + 2\text{HCl}(aq)
ightarrow \text{CoCl}_2(aq) + \text{H}_2(g) \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 2\text{CO}(g) + \text{O}_2(g)
ightarrow 2\text{CO}_2(g) \)