QUESTION IMAGE
Question
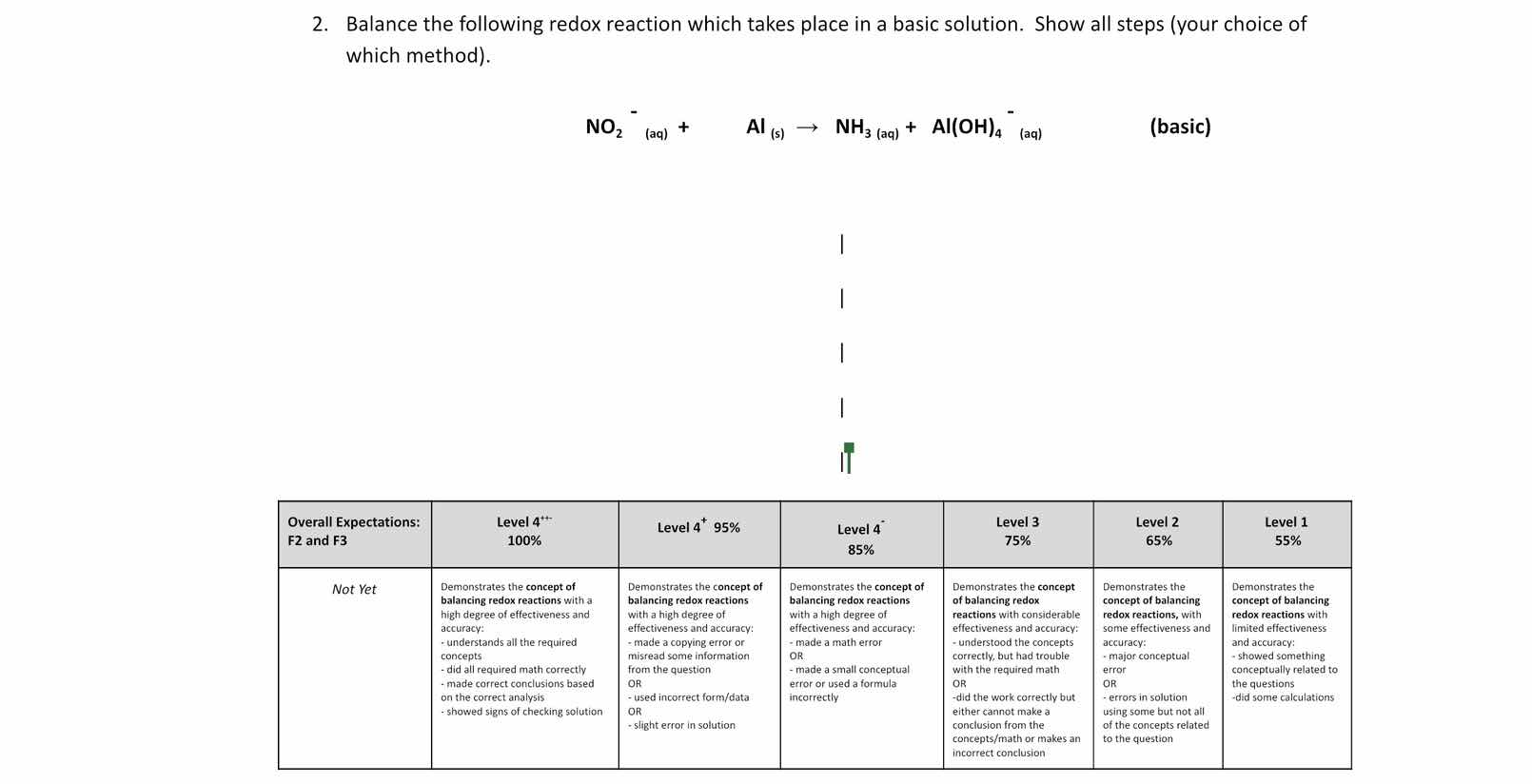
- balance the following redox reaction which takes place in a basic solution. show all steps (your choice of which method).
\\(\text{no}_2^-\\) (aq) + \\(\text{al}_{(s)}\\) \\(\
ightarrow\\) \\(\text{nh}_3\\) (aq) + \\(\text{al(oh)}_4^-\\) (aq) (basic)
overall expectations:
f2 and f3\tlevel 4++ 100%\tlevel 4+ 95%\tlevel 4- 85%\tlevel 3 75%\tlevel 2 65%\tlevel 1 55%
not yet\tdemonstrates the concept of balancing redox reactions with a high degree of effectiveness and accuracy:
- understands all the required concepts
- did all required math correctly
- made correct conclusions based on the correct analysis
- showed signs of checking solution\tdemonstrates the concept of balancing redox reactions with a high degree of effectiveness and accuracy:
- made a copying error or misread some information from the question
or
- used incorrect form/data
or
- slight error in solution\tdemonstrates the concept of balancing redox reactions with a high degree of effectiveness and accuracy:
- made a math error
or
- made a small conceptual error or used a formula incorrectly\tdemonstrates the concept of balancing redox reactions with considerable effectiveness and accuracy:
- understood the concepts correctly, but had trouble with the required math
or
- did the work correctly but either cannot make a conclusion from the concepts/math or makes an incorrect conclusion\tdemonstrates the concept of balancing redox reactions with some effectiveness and accuracy:
- major conceptual error
- errors in solution using some but not all of the concepts related to the question\tdemonstrates the concept of balancing redox reactions with limited effectiveness and accuracy:
- showed something conceptually related to the questions
- did some calculations
Step1: Identify Oxidation and Reduction Half - Reactions
- Oxidation (Al to \(Al(OH)_4^-\)):
- The oxidation state of Al in \(Al(s)\) is 0, and in \(Al(OH)_4^-\), let the oxidation state of Al be \(x\). We know that the oxidation state of O is - 2 and H is + 1. So, \(x+4\times(- 2)+4\times(+1)= - 1\), which gives \(x - 8 + 4=-1\), and \(x = + 3\). So Al is oxidized (oxidation state increases from 0 to + 3).
- The half - reaction for oxidation: \(Al(s)\to Al(OH)_4^-(aq)\)
- Reduction (\(NO_2^-\) to \(NH_3\)):
- The oxidation state of N in \(NO_2^-\): Let the oxidation state of N be \(y\). Then \(y+2\times(-2)=-1\), so \(y - 4=-1\), and \(y = + 3\). In \(NH_3\), the oxidation state of N is - 3 (since H is + 1, \(x + 3\times(+1)=0\), \(x=-3\)). So N is reduced (oxidation state decreases from + 3 to - 3).
- The half - reaction for reduction: \(NO_2^-(aq)\to NH_3(aq)\)
Step2: Balance Atoms Other Than O and H in Each Half - Reaction
- Oxidation half - reaction:
- Al atoms are already balanced: \(Al(s)\to Al(OH)_4^-(aq)\)
- Reduction half - reaction:
- N atoms are already balanced: \(NO_2^-(aq)\to NH_3(aq)\)
Step3: Balance O Atoms by Adding \(H_2O\)
- Oxidation half - reaction:
- There are 4 O atoms on the right. So we add 4 \(H_2O\) to the left: \(Al(s)+4H_2O(l)\to Al(OH)_4^-(aq)\)
- Reduction half - reaction:
- There are 2 O atoms on the left. So we add 2 \(H_2O\) to the right: \(NO_2^-(aq)\to NH_3(aq)+2H_2O(l)\)
Step4: Balance H Atoms by Adding \(H^+\) (then adjust for basic solution later)
- Oxidation half - reaction:
- On the left, we have 8 H atoms from \(4H_2O\). On the right, we have 4 H atoms in \(Al(OH)_4^-\). So we add 4 \(H^+\) to the right: \(Al(s)+4H_2O(l)\to Al(OH)_4^-(aq)+4H^+(aq)\)
- Reduction half - reaction:
- On the right, we have 3 H atoms in \(NH_3\) and 4 H atoms from \(2H_2O\), a total of 7 H atoms. So we add 7 \(H^+\) to the left: \(NO_2^-(aq)+7H^+(aq)\to NH_3(aq)+2H_2O(l)\)
Step5: Balance Charge by Adding \(e^-\)
- Oxidation half - reaction:
- Left side charge: 0 (Al is neutral, \(H_2O\) is neutral). Right side charge: \(-1 + 4\times(+1)=+3\). So we need to lose 3 electrons (oxidation is loss of electrons) to balance the charge: \(Al(s)+4H_2O(l)\to Al(OH)_4^-(aq)+4H^+(aq)+3e^-\)
- Reduction half - reaction:
- Left side charge: \(-1+7\times(+1)=+6\). Right side charge: 0 ( \(NH_3\) and \(H_2O\) are neutral). So we need to gain 6 electrons (reduction is gain of electrons) to balance the charge: \(NO_2^-(aq)+7H^+(aq)+6e^-\to NH_3(aq)+2H_2O(l)\)
Step6: Equalize the Number of Electrons Gained and Lost
- The oxidation half - reaction loses 3 \(e^-\) and the reduction half - reaction gains 6 \(e^-\). Multiply the oxidation half - reaction by 2 to make the number of electrons equal (6 electrons lost and 6 electrons gained):
- Oxidation (after multiplication): \(2Al(s)+8H_2O(l)\to 2Al(OH)_4^-(aq)+8H^+(aq)+6e^-\)
- Reduction (unchanged): \(NO_2^-(aq)+7H^+(aq)+6e^-\to NH_3(aq)+2H_2O(l)\)
Step7: Add the Two Half - Reactions Together
- Add the two half - reactions:
\(2Al(s)+8H_2O(l)+NO_2^-(aq)+7H^+(aq)+6e^-\to 2Al(OH)_4^-(aq)+8H^+(aq)+6e^-+NH_3(aq)+2H_2O(l)\)
- Cancel out the electrons (\(6e^-\) on both sides), and simplify the \(H^+\) and \(H_2O\) terms:
- For \(H_2O\): \(8H_2O - 2H_2O = 6H_2O\)
- For \(H^+\): \(7H^+-8H^+=-H^+\)
- The equation becomes: \(2Al(s)+NO_2^-(aq)+6H_2O(l)\to 2Al(OH)_4^-(aq)+NH_3(aq)-H^+(aq)\)
Step8: Adjust for Basic Solution (Add \(OH^-\) to Neutralize \(H^+\))
- Since the solution is basic, we add…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Step1: Identify Oxidation and Reduction Half - Reactions
- Oxidation (Al to \(Al(OH)_4^-\)):
- The oxidation state of Al in \(Al(s)\) is 0, and in \(Al(OH)_4^-\), let the oxidation state of Al be \(x\). We know that the oxidation state of O is - 2 and H is + 1. So, \(x+4\times(- 2)+4\times(+1)= - 1\), which gives \(x - 8 + 4=-1\), and \(x = + 3\). So Al is oxidized (oxidation state increases from 0 to + 3).
- The half - reaction for oxidation: \(Al(s)\to Al(OH)_4^-(aq)\)
- Reduction (\(NO_2^-\) to \(NH_3\)):
- The oxidation state of N in \(NO_2^-\): Let the oxidation state of N be \(y\). Then \(y+2\times(-2)=-1\), so \(y - 4=-1\), and \(y = + 3\). In \(NH_3\), the oxidation state of N is - 3 (since H is + 1, \(x + 3\times(+1)=0\), \(x=-3\)). So N is reduced (oxidation state decreases from + 3 to - 3).
- The half - reaction for reduction: \(NO_2^-(aq)\to NH_3(aq)\)
Step2: Balance Atoms Other Than O and H in Each Half - Reaction
- Oxidation half - reaction:
- Al atoms are already balanced: \(Al(s)\to Al(OH)_4^-(aq)\)
- Reduction half - reaction:
- N atoms are already balanced: \(NO_2^-(aq)\to NH_3(aq)\)
Step3: Balance O Atoms by Adding \(H_2O\)
- Oxidation half - reaction:
- There are 4 O atoms on the right. So we add 4 \(H_2O\) to the left: \(Al(s)+4H_2O(l)\to Al(OH)_4^-(aq)\)
- Reduction half - reaction:
- There are 2 O atoms on the left. So we add 2 \(H_2O\) to the right: \(NO_2^-(aq)\to NH_3(aq)+2H_2O(l)\)
Step4: Balance H Atoms by Adding \(H^+\) (then adjust for basic solution later)
- Oxidation half - reaction:
- On the left, we have 8 H atoms from \(4H_2O\). On the right, we have 4 H atoms in \(Al(OH)_4^-\). So we add 4 \(H^+\) to the right: \(Al(s)+4H_2O(l)\to Al(OH)_4^-(aq)+4H^+(aq)\)
- Reduction half - reaction:
- On the right, we have 3 H atoms in \(NH_3\) and 4 H atoms from \(2H_2O\), a total of 7 H atoms. So we add 7 \(H^+\) to the left: \(NO_2^-(aq)+7H^+(aq)\to NH_3(aq)+2H_2O(l)\)
Step5: Balance Charge by Adding \(e^-\)
- Oxidation half - reaction:
- Left side charge: 0 (Al is neutral, \(H_2O\) is neutral). Right side charge: \(-1 + 4\times(+1)=+3\). So we need to lose 3 electrons (oxidation is loss of electrons) to balance the charge: \(Al(s)+4H_2O(l)\to Al(OH)_4^-(aq)+4H^+(aq)+3e^-\)
- Reduction half - reaction:
- Left side charge: \(-1+7\times(+1)=+6\). Right side charge: 0 ( \(NH_3\) and \(H_2O\) are neutral). So we need to gain 6 electrons (reduction is gain of electrons) to balance the charge: \(NO_2^-(aq)+7H^+(aq)+6e^-\to NH_3(aq)+2H_2O(l)\)
Step6: Equalize the Number of Electrons Gained and Lost
- The oxidation half - reaction loses 3 \(e^-\) and the reduction half - reaction gains 6 \(e^-\). Multiply the oxidation half - reaction by 2 to make the number of electrons equal (6 electrons lost and 6 electrons gained):
- Oxidation (after multiplication): \(2Al(s)+8H_2O(l)\to 2Al(OH)_4^-(aq)+8H^+(aq)+6e^-\)
- Reduction (unchanged): \(NO_2^-(aq)+7H^+(aq)+6e^-\to NH_3(aq)+2H_2O(l)\)
Step7: Add the Two Half - Reactions Together
- Add the two half - reactions:
\(2Al(s)+8H_2O(l)+NO_2^-(aq)+7H^+(aq)+6e^-\to 2Al(OH)_4^-(aq)+8H^+(aq)+6e^-+NH_3(aq)+2H_2O(l)\)
- Cancel out the electrons (\(6e^-\) on both sides), and simplify the \(H^+\) and \(H_2O\) terms:
- For \(H_2O\): \(8H_2O - 2H_2O = 6H_2O\)
- For \(H^+\): \(7H^+-8H^+=-H^+\)
- The equation becomes: \(2Al(s)+NO_2^-(aq)+6H_2O(l)\to 2Al(OH)_4^-(aq)+NH_3(aq)-H^+(aq)\)
Step8: Adjust for Basic Solution (Add \(OH^-\) to Neutralize \(H^+\))
- Since the solution is basic, we add \(OH^-\) to both sides to neutralize \(H^+\). We have \(-H^+\) on the right, which is equivalent to \(H^+\) on the left. So we add 1 \(OH^-\) to both sides:
\(2Al(s)+NO_2^-(aq)+6H_2O(l)+OH^-(aq)\to 2Al(OH)_4^-(aq)+NH_3(aq)-H^+(aq)+OH^-(aq)\)
- Since \(H^+(aq)+OH^-(aq)=H_2O(l)\), the right - hand side becomes \(NH_3(aq)+H_2O(l)\) and the left - hand side has \(6H_2O + H_2O\) (from the reaction of \(H^+\) and \(OH^-\))? Wait, no. Let's re - do this step. The equation after step 7 is \(2Al(s)+NO_2^-(aq)+6H_2O(l)\to 2Al(OH)_4^-(aq)+NH_3(aq)+H^+(aq)\) (I made a sign error earlier. The correct simplification of \(7H^+-8H^+\) is \(-H^+\), but when we move \(H^+\) to the right, it's \(+H^+\) on the right. So the equation is \(2Al(s)+NO_2^-(aq)+6H_2O(l)\to 2Al(OH)_4^-(aq)+NH_3(aq)+H^+(aq)\))
- Now, to neutralize \(H^+\) in basic solution, we add \(OH^-\) to both sides:
\(2Al(s)+NO_2^-(aq)+6H_2O(l)+OH^-(aq)\to 2Al(OH)_4^-(aq)+NH_3(aq)+H^+(aq)+OH^-(aq)\)
- Since \(H^+(aq)+OH^-(aq)=H_2O(l)\), the right - hand side becomes \(NH_3(aq)+H_2O(l)\) and the left - hand side has \(6H_2O+OH^-\). Wait, a better way: In basic solution, we can also balance H and O by using \(OH^-\) and \(H_2O\) from the start. Let's try an alternative approach for balancing in basic solution (to correct the earlier mistake).
Alternative Step for Basic Solution Balancing (Starting from Step 3)
- Oxidation half - reaction (basic solution):
- \(Al(s)\to Al(OH)_4^-(aq)\)
- Balance O: Add 4 \(OH^-\) to the left (since in basic solution, we use \(OH^-\) and \(H_2O\)): \(Al(s)+4OH^-(aq)\to Al(OH)_4^-(aq)\)
- Balance H: The left has 4 \(H\) from \(4OH^-\), the right has 4 \(H\) in \(Al(OH)_4^-\), so H is balanced.
- Balance charge: Left charge: \(0 + 4\times(-1)=-4\), Right charge: \(-1\). So we need to lose 3 electrons (oxidation): \(Al(s)+4OH^-(aq)\to Al(OH)_4^-(aq)+3e^-\)
- Reduction half - reaction (basic solution):
- \(NO_2^-(aq)\to NH_3(aq)\)
- Balance O: Add 1 \(H_2O\) to the right (to balance 2 O on left): \(NO_2^-(aq)\to NH_3(aq)+2H_2O(l)\)
- Balance H: Add 5 \(H_2O\) to the left and 5 \(OH^-\) to the right (in basic solution, to balance H, we use \(H_2O\) and \(OH^-\); the number of H atoms: left needs 5 more H (since \(NH_3\) has 3 H and \(2H_2O\) has 4 H, total 7 H on right, and \(NO_2^-\) has 0 H on left. Wait, better formula: For basic solution, to balance H, if we have \(a\) H on right and \(b\) H on left, we add \(\frac{a - b}{2}\) \(H_2O\) to the side with less H and \(\frac{a - b}{2}\) \(OH^-\) to the other side.
- Let's do it properly:
- \(NO_2^-(aq)\to NH_3(aq)\)
- Balance O: \(NO_2^-(aq)\to NH_3(aq)+2H_2O(l)\) (2 O on left, 2 O on right)
- Balance H: Right has \(3 + 4 = 7\) H, left has 0 H. So we add 7 \(H_2O\) to the left and 7 \(OH^-\) to the right: \(NO_2^-(aq)+7H_2O(l)\to NH_3(aq)+2H_2O(l)+7OH^-(aq)\)
- Simplify \(H_2O\): \(NO_2^-(aq)+5H_2O(l)\to NH_3(aq)+7OH^-(aq)\)
- Balance charge: Left charge: \(-1+0=-1\), Right charge: \(0 + 7\times(-1)=-7\). So we need to gain 6 electrons (reduction): \(NO_2^-(aq)+5H_2O(l)+6e^-\to NH_3(aq)+7OH^-(aq)\)
- Now, the oxidation half - reaction (from alternative step) is \(Al(s)+4OH^-(aq)\to Al(OH)_4^-(aq)+3e^-\) (multiply by 2 to get 6 electrons lost): \(2Al(s)+8OH^-(aq)\to 2Al(OH)_4^-(aq)+6e^-\)
- The reduction half - reaction is \(NO_2^-(aq)+5H_2O(l)+6e^-\to NH_3(aq)+7OH^-(aq)\)
- Add the two half - reactions:
\(2Al(s)+8OH^-(aq)+NO_2^-(aq)+5H_2O(l)+6e^-\to 2Al(OH)_4^-(aq)+6e^-+NH_3(aq)+7OH^-(aq)\)
- Cancel electrons, simplify \(OH^-\) and \(H_2O\):
- \(OH^-\): \(8OH^--7OH^- = OH^-\)
- \(H_2O\): 5 \(H_2O\)
- The equation becomes: \(2Al(s)+NO_2^-(aq)+5H_2O(l)+OH^-(aq)\to 2Al(OH)_4^-(aq)+NH_3(aq)\)
- Or, we can write it as: \(2Al(s)+NO_2^-(aq)+6H_2O(l)\to 2Al(OH)_4^-(aq)+NH_3(aq)+OH^-(aq)\) (by adding \(H_2O\) to both sides to make the \(H_2O\) on left as 6, since \(5H_2O + H_2O=6H_2O\) and \(OH^-+H_2O = H_2O+OH^-\), but this is equivalent to the previous equation).
- Let's check the final balanced equation:
- Atoms:
- Al: 2 on left, 2 on right.
- N: 1 on left, 1 on right.
- O: Left: \(1\times2 + 6\times1=8\); Right: \(2\times4+1\times1 = 9\)? Wait, no, in \(2Al(OH)_4^-\) there are \(2\times4 = 8\) O, in \(NH_3\) 0, in \(OH^-\) 1, total 9. On left: \(NO_2^-\) has 2 O, \(6H_2O\) has 6 O, total 8 O. Wait, there is a mistake. Let's re - balance the reduction half - reaction correctly.
Correct Reduction Half - Reaction Balancing in Basic Solution
- \(NO_2^-(aq)\to NH_3(aq)\)
- Balance N: 1 N on each side.
- Balance O: Add 1 \(H_2O\) to the right (2 O on left, 2 O in \(H_2O\) on right): \(NO_2^-(aq)\to NH_3(aq)+2H_2O(l)\)
- Balance H: In basic solution, we add \(OH^-\) to the left and \(H_2O\) to the right. The number of H atoms: right has \(3 + 4 = 7\) H (3 from \(NH_3\), 4 from \(2H_2O\)). So we add 7 \(H_2O\) to the left and 7 \(OH^-\) to the right: \(NO_2^-(aq)+7H_2O(l)\to NH_3(aq)+2H_2O(l)+7OH^-(aq)\)
- Simplify \(H_2O\): \(NO_2^-(aq)+5H_2O(l)\to NH_3(aq)+7OH^-(aq)\)
- Balance charge: Left charge: \(-1\), Right charge: \(0+7\times(-1)=-7\). So we need to gain 6 electrons: \(NO_2^-(aq)+5H_2O(l)+6e^-\to NH_3(aq)+7OH^-(aq)\) (charge on left: \(-1 + 0=-1\), on right: \(0+7\times(-1)+6\times(0)=-7\)? Wait, no, electrons have charge - 1. So left charge: \(-1+6\times(-1)=-7\), right charge: \(-7\). Yes, that's correct. So the reduction half - reaction charge balance: \(NO_2^-(aq)+5H_2O(l)+6e^-\) has charge \(-1+0 + 6\times(-1)=-7\), and \(NH_3(aq)+7OH^-(aq)\) has charge \(0+7\times(-1)=-7\).
- Oxidation half - reaction (basic): \(Al(s)\to Al(OH)_4^-(aq)\)
- Balance O: Add 4 \(OH^-\) to left: \(Al(s)+4OH^-(aq)\to Al(OH)_4^-(aq)\)
- Balance charge: Left: \(0+4\times(-1)=-4\), Right: \(-1\). So lose 3 electrons: \(Al(s)+4OH^-(aq)\to Al(OH)_4^-(aq)+3e^-\) (charge: left \(-4\), right \(-1+3\times(-1)=-4\))
- Multiply oxidation half - reaction by 2: \(2Al(s)+8OH^-(aq)\to 2Al(OH)_4^-(aq)+6e^-\) (charge: left \(8\times(-1)+0=-8\), right \(2\times(-1)+6\times(-1)=-8\))
- Add reduction and oxidized half - reactions: