QUESTION IMAGE
Question
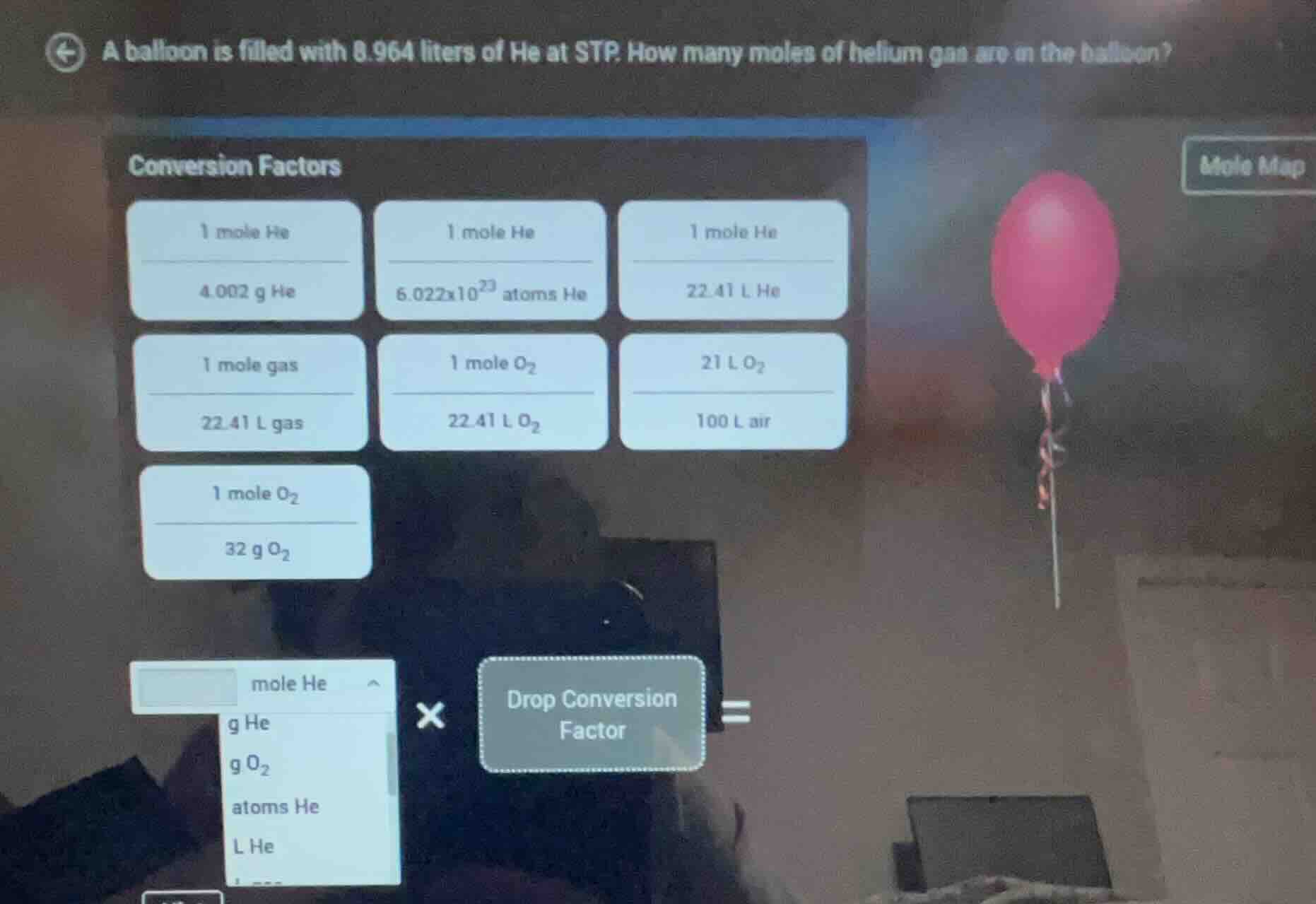
a balloon is filled with 8.964 liters of he at stp. how many moles of helium gas are in the balloon?
conversion factors
1 mole he / 4.002 g he
1 mole he / 6.022×10²³ atoms he
1 mole he / 22.41 l he
1 mole gas / 22.41 l gas
1 mole o₂ / 22.41 l o₂
21 l o₂ / 100 l air
1 mole o₂ / 32 g o₂
(options for unit conversion: mole he, g he, g o₂, atoms he, l he)
Step1: Identify the conversion factor
We know that at STP, 1 mole of any gas occupies 22.41 L. For helium, the conversion factor is $\frac{1\ \text{mole He}}{22.41\ \text{L He}}$.
Step2: Set up the calculation
We have 8.964 L of He. To find the moles, we multiply the volume by the conversion factor:
$8.964\ \text{L He} \times \frac{1\ \text{mole He}}{22.41\ \text{L He}}$
Step3: Perform the division
Calculate $8.964\div22.41 = 0.4$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
0.4 moles