QUESTION IMAGE
Question
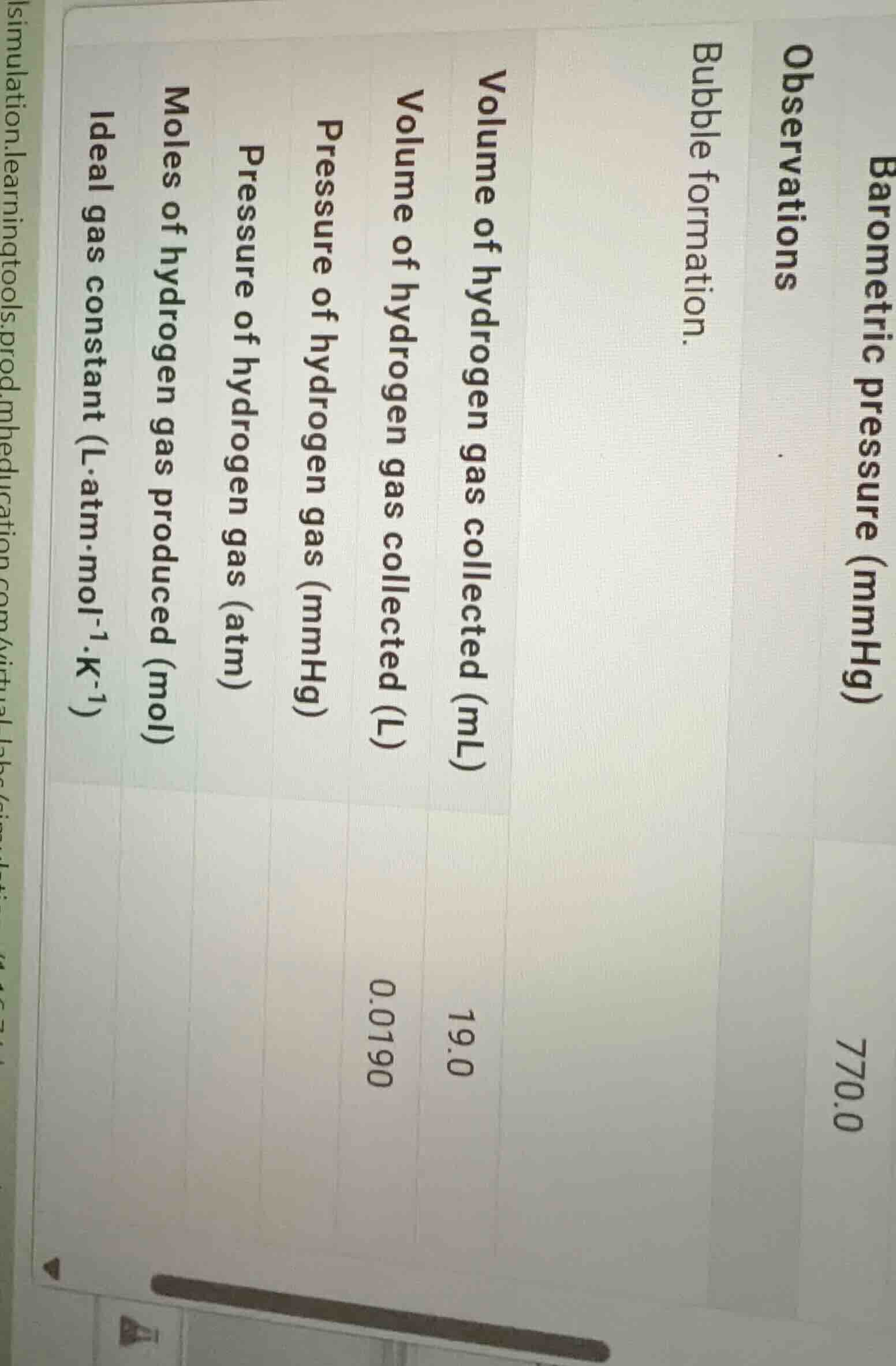
barometric pressure (mmhg)
observations
bubble formation.
volume of hydrogen gas collected (ml)
volume of hydrogen gas collected (l)
pressure of hydrogen gas (mmhg)
pressure of hydrogen gas (atm)
moles of hydrogen gas produced (mol)
ideal gas constant (l·atm·mol⁻¹·k⁻¹)
770.0
19.0
0.0190
To solve for the moles of hydrogen gas produced, we use the ideal gas law, \( PV = nRT \), where:
- \( P \) = pressure of hydrogen gas (atm),
- \( V \) = volume of hydrogen gas (L),
- \( n \) = moles of gas,
- \( R \) = ideal gas constant (\( 0.0821 \, \text{L·atm·mol}^{-1}·\text{K}^{-1} \)),
- \( T \) (not given, but we assume room temperature \( T = 298 \, \text{K} \) for estimation, or we can solve symbolically if \( T \) is provided).
Step 1: Convert pressure to atm
Barometric pressure = \( 770.0 \, \text{mmHg} \). Assume the pressure of \( \text{H}_2 \) is equal to barometric pressure (if no vapor pressure correction is needed).
\( 1 \, \text{atm} = 760 \, \text{mmHg} \), so:
\( P = \frac{770.0 \, \text{mmHg}}{760 \, \text{mmHg/atm}} \approx 1.013 \, \text{atm} \).
Step 2: Convert volume to liters
Volume of \( \text{H}_2 \) = \( 19.0 \, \text{mL} = 0.0190 \, \text{L} \) (wait, the table shows "Volume of hydrogen gas collected (L)" as \( 0.0190 \, \text{L} \)? Wait, the table has:
- Volume of hydrogen gas collected (mL): \( 19.0 \)
- Volume of hydrogen gas collected (L): \( 0.0190 \) (since \( 19.0 \, \text{mL} = 0.0190 \, \text{L} \), correct).
Step 3: Rearrange ideal gas law to solve for \( n \)
From \( PV = nRT \), solve for \( n \):
\( n = \frac{PV}{RT} \).
Step 4: Substitute values (assuming \( T = 298 \, \text{K} \))
\( P = 1.013 \, \text{atm} \), \( V = 0.0190 \, \text{L} \), \( R = 0.0821 \, \text{L·atm·mol}^{-1}·\text{K}^{-1} \), \( T = 298 \, \text{K} \):
\( n = \frac{(1.013 \, \text{atm})(0.0190 \, \text{L})}{(0.0821 \, \text{L·atm·mol}^{-1}·\text{K}^{-1})(298 \, \text{K})} \)
Calculate numerator: \( (1.013)(0.0190) \approx 0.01925 \)
Calculate denominator: \( (0.0821)(298) \approx 24.47 \)
\( n \approx \frac{0.01925}{24.47} \approx 0.000787 \, \text{mol} \).
Wait, but the table already has "Moles of hydrogen gas produced (mol)" as a blank. If we use the given \( V = 0.0190 \, \text{L} \), \( P = 1.013 \, \text{atm} \), \( R = 0.0821 \), \( T = 298 \, \text{K} \), the calculation gives \( n \approx 0.0008 \, \text{mol} \).
If we instead use the values directly (e.g., \( V = 0.0190 \, \text{L} \), \( P = 1.013 \, \text{atm} \), \( T = 298 \, \text{K} \)):
\( n = \frac{(1.013)(0.0190)}{(0.0821)(298)} \approx 7.9 \times 10^{-4} \, \text{mol} \) (or \( 0.00079 \, \text{mol} \)).
Final Answer
The moles of hydrogen gas produced is approximately \( \boldsymbol{7.9 \times 10^{-4} \, \text{mol}} \) (or \( 0.00079 \, \text{mol} \)) (depending on \( T \); if \( T \) is different, recalculate).
(Note: If \( T \) is provided, substitute it for more accuracy. If the problem assumes \( T = 273 \, \text{K} \) (STP), then \( T = 273 \, \text{K} \), and:
\( n = \frac{(1.013)(0.0190)}{(0.0821)(273)} \approx 0.00086 \, \text{mol} \).)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve for the moles of hydrogen gas produced, we use the ideal gas law, \( PV = nRT \), where:
- \( P \) = pressure of hydrogen gas (atm),
- \( V \) = volume of hydrogen gas (L),
- \( n \) = moles of gas,
- \( R \) = ideal gas constant (\( 0.0821 \, \text{L·atm·mol}^{-1}·\text{K}^{-1} \)),
- \( T \) (not given, but we assume room temperature \( T = 298 \, \text{K} \) for estimation, or we can solve symbolically if \( T \) is provided).
Step 1: Convert pressure to atm
Barometric pressure = \( 770.0 \, \text{mmHg} \). Assume the pressure of \( \text{H}_2 \) is equal to barometric pressure (if no vapor pressure correction is needed).
\( 1 \, \text{atm} = 760 \, \text{mmHg} \), so:
\( P = \frac{770.0 \, \text{mmHg}}{760 \, \text{mmHg/atm}} \approx 1.013 \, \text{atm} \).
Step 2: Convert volume to liters
Volume of \( \text{H}_2 \) = \( 19.0 \, \text{mL} = 0.0190 \, \text{L} \) (wait, the table shows "Volume of hydrogen gas collected (L)" as \( 0.0190 \, \text{L} \)? Wait, the table has:
- Volume of hydrogen gas collected (mL): \( 19.0 \)
- Volume of hydrogen gas collected (L): \( 0.0190 \) (since \( 19.0 \, \text{mL} = 0.0190 \, \text{L} \), correct).
Step 3: Rearrange ideal gas law to solve for \( n \)
From \( PV = nRT \), solve for \( n \):
\( n = \frac{PV}{RT} \).
Step 4: Substitute values (assuming \( T = 298 \, \text{K} \))
\( P = 1.013 \, \text{atm} \), \( V = 0.0190 \, \text{L} \), \( R = 0.0821 \, \text{L·atm·mol}^{-1}·\text{K}^{-1} \), \( T = 298 \, \text{K} \):
\( n = \frac{(1.013 \, \text{atm})(0.0190 \, \text{L})}{(0.0821 \, \text{L·atm·mol}^{-1}·\text{K}^{-1})(298 \, \text{K})} \)
Calculate numerator: \( (1.013)(0.0190) \approx 0.01925 \)
Calculate denominator: \( (0.0821)(298) \approx 24.47 \)
\( n \approx \frac{0.01925}{24.47} \approx 0.000787 \, \text{mol} \).
Wait, but the table already has "Moles of hydrogen gas produced (mol)" as a blank. If we use the given \( V = 0.0190 \, \text{L} \), \( P = 1.013 \, \text{atm} \), \( R = 0.0821 \), \( T = 298 \, \text{K} \), the calculation gives \( n \approx 0.0008 \, \text{mol} \).
If we instead use the values directly (e.g., \( V = 0.0190 \, \text{L} \), \( P = 1.013 \, \text{atm} \), \( T = 298 \, \text{K} \)):
\( n = \frac{(1.013)(0.0190)}{(0.0821)(298)} \approx 7.9 \times 10^{-4} \, \text{mol} \) (or \( 0.00079 \, \text{mol} \)).
Final Answer
The moles of hydrogen gas produced is approximately \( \boldsymbol{7.9 \times 10^{-4} \, \text{mol}} \) (or \( 0.00079 \, \text{mol} \)) (depending on \( T \); if \( T \) is different, recalculate).
(Note: If \( T \) is provided, substitute it for more accuracy. If the problem assumes \( T = 273 \, \text{K} \) (STP), then \( T = 273 \, \text{K} \), and:
\( n = \frac{(1.013)(0.0190)}{(0.0821)(273)} \approx 0.00086 \, \text{mol} \).)