QUESTION IMAGE
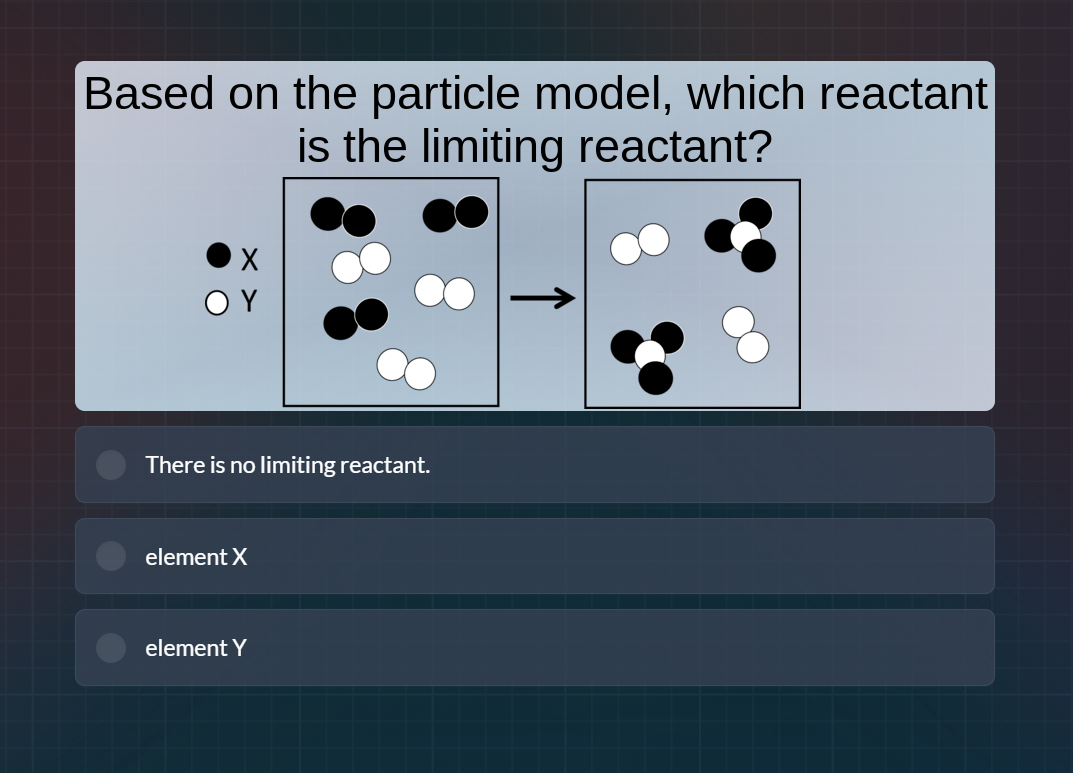
Question
based on the particle model, which reactant is the limiting reactant?
x (black circle), y (white circle)
left box (reactants): 3 pairs of black circles (x₂), 3 pairs of white circles (y₂)
right box (products): 2 molecules with 1 white and 2 black (y x₂), 1 pair of white (y₂), 1 pair of white (y₂)
options:
there is no limiting reactant.
element x
element y
- First, count the number of X (black) and Y (white) molecules initially.
- X molecules: Each black pair is \(X_2\). There are 3 pairs, so \(3\times2 = 6\) X atoms (or 3 \(X_2\) molecules).
- Y molecules: Each white pair is \(Y_2\). There are 3 pairs, so \(3\times2 = 6\) Y atoms (or 3 \(Y_2\) molecules).
- Analyze the product: The product has \(XY_2\) (each has 1 X and 2 Y) and leftover \(Y_2\).
- For \(XY_2\) formation, the ratio of \(X_2\) to \(Y_2\) should be such that 1 \(X_2\) reacts with 2 \(Y_2\) (since \(X_2 + 2Y_2
ightarrow2XY_2\))? Wait, no, looking at the product: each \(XY_2\) has 1 X and 2 Y. Wait, actually, from the reactants: \(X_2\) (black pairs) and \(Y_2\) (white pairs). Let's count the product:
- Number of \(XY_2\) molecules: 2 (each with 1 X and 2 Y). Wait, no, the product has two \(XY_2\) (each with 1 X and 2 Y) and one leftover \(Y_2\) (white pair). Wait, initial \(Y_2\): 3 pairs (6 Y atoms). Initial \(X_2\): 3 pairs (6 X atoms).
- But after reaction, there is leftover \(Y_2\), which means X was used up first? Wait, no, wait the product: the \(XY_2\) molecules: each has 1 X and 2 Y. Let's count the number of X used: in the product, there are 2 \(XY_2\) molecules? Wait, no, looking at the diagram:
- Reactants: 3 \(X_2\) (black pairs: 3 groups of 2 black circles) and 3 \(Y_2\) (white pairs: 3 groups of 2 white circles).
- Products: 2 \(XY_2\) (each with 1 black and 2 white) and 1 \(Y_2\) (white pair) and 1 \(Y_2\)? Wait, no, the right box has: two \(XY_2\) (each with 1 X and 2 Y), one \(Y_2\) (white pair), and one \(Y_2\)? Wait, no, the white circles: one pair (2 white) and the \(XY_2\) have 2 white each. Wait, maybe the reaction is \(X_2 + Y_2
ightarrow XY_2\) (but stoichiometry: 1 \(X_2\) reacts with 1 \(Y_2\) to make 2 \(XY_2\)? No, that doesn't fit. Wait, maybe the correct ratio is \(X_2 + 2Y_2
ightarrow 2XY_2\). Let's check:
- If we have 3 \(X_2\) (6 X atoms) and 3 \(Y_2\) (6 Y atoms). For the reaction \(X_2 + 2Y_2
ightarrow 2XY_2\), each \(X_2\) needs 2 \(Y_2\). So 3 \(X_2\) would need 6 \(Y_2\), but we only have 3 \(Y_2\). Wait, that can't be. Wait, maybe the reaction is \(X_2 + Y_2
ightarrow 2XY\), but no, the product is \(XY_2\). Wait, maybe I made a mistake. Let's count the number of X and Y in products:
- Products:
- \(XY_2\) molecules: 2 (each with 1 X and 2 Y) → 2 X atoms used, 4 Y atoms used.
- Leftover \(Y_2\): 1 pair (2 Y atoms) → 2 Y atoms leftover.
- Total Y atoms: 4 + 2 = 6 (matches initial 3 \(Y_2\) (6 Y atoms)).
- X atoms used: 2 (from 2 \(XY_2\) molecules) → initial X atoms: 6 (from 3 \(X_2\) molecules). Wait, that means X is in excess? No, that can't be. Wait, maybe the reaction is \(X + 2Y
ightarrow XY_2\), but the reactants are \(X_2\) and \(Y_2\). So \(X_2\) dissociates into 2 X, \(Y_2\) dissociates into 2 Y. Then 2 X + 4 Y → 2 \(XY_2\). So for each \(XY_2\), we need 1 X and 2 Y.
- Initial X: 6 (3 \(X_2\) → 3×2=6 X). Initial Y: 6 (3 \(Y_2\) → 3×2=6 Y).
- In products, we have 2 \(XY_2\) (using 2 X and 4 Y) and leftover Y: 6 - 4 = 2 Y (which is 1 \(Y_2\)). So X used: 2, X leftover: 6 - 2 = 4? No, that's not right. Wait, maybe the reactants are \(X_2\) (black pairs) and \(Y_2\) (white pairs), and the reaction is \(X_2 + 2Y_2
ightarrow 2XY_2\). So 1 \(X_2\) reacts with 2 \(Y_2\) to make 2 \(XY_2\).
- Initial \(X_2\): 3, initial \(Y_2\): 3.
- If we use 1 \(X_2\), we need 2 \(Y_2\). So with 3 \(X_2\), we would need 6 \(Y_2\), but we only have 3 \(Y_2\). So actually, \(Y_2\) is limiting? No, that contradicts. Wait, maybe I got the reac…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
element X