QUESTION IMAGE
Question
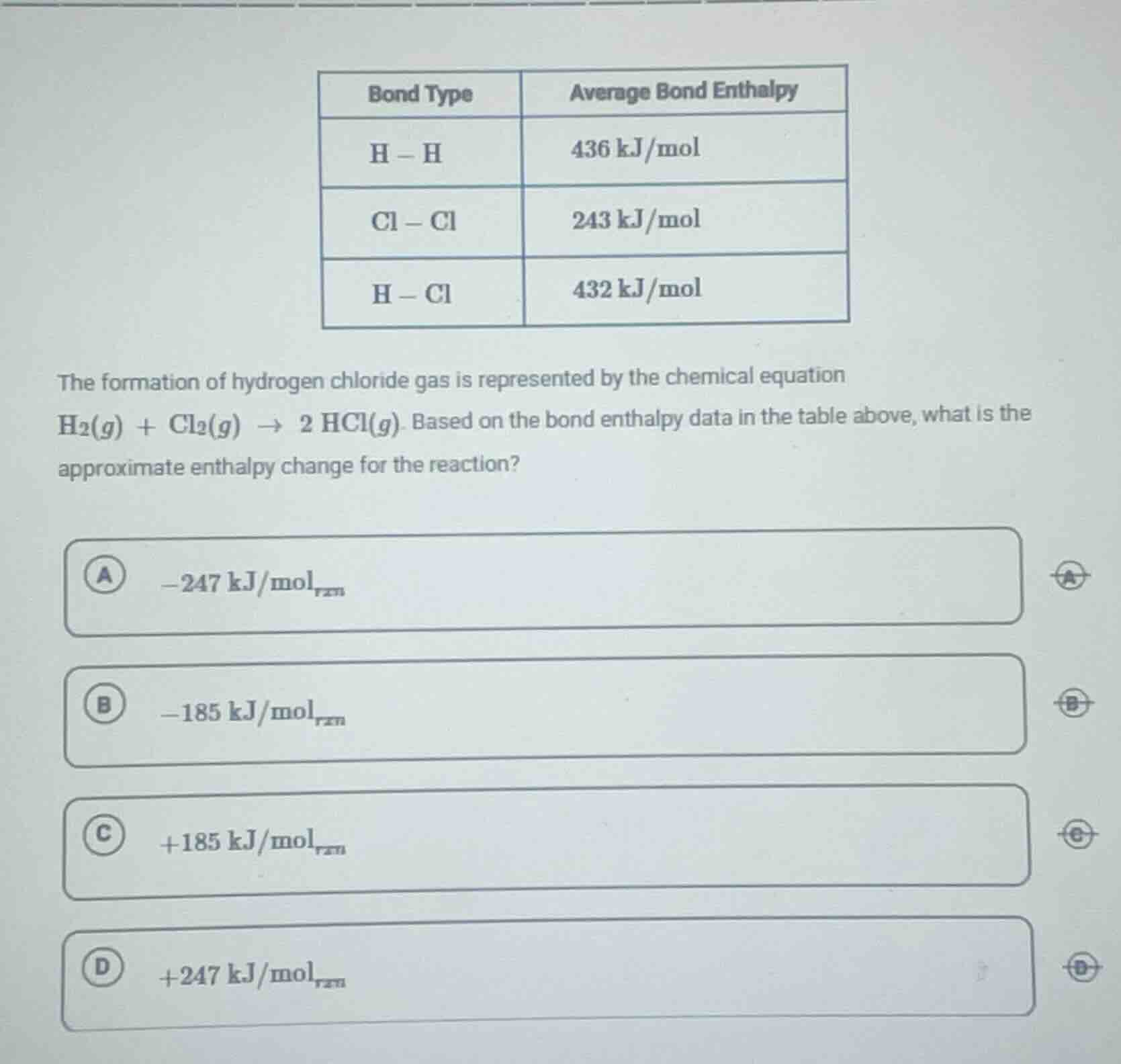
bond type | average bond enthalpy
h - h | 436 kj/mol
cl - cl | 243 kj/mol
h - cl | 432 kj/mol
the formation of hydrogen chloride gas is represented by the chemical equation $ce{h2(g) + cl2(g) -> 2 hcl(g)}$. based on the bond enthalpy data in the table above, what is the approximate enthalpy change for the reaction?
a $-247\\ \mathrm{kj/mol_{rxn}}$
b $-185\\ \mathrm{kj/mol_{rxn}}$
c $+185\\ \mathrm{kj/mol_{rxn}}$
d $+247\\ \mathrm{kj/mol_{rxn}}$
Step1: Recall the formula for enthalpy change using bond enthalpies
The enthalpy change ($\Delta H$) of a reaction is calculated as the sum of the bond enthalpies of the bonds broken minus the sum of the bond enthalpies of the bonds formed. Mathematically, $\Delta H = \sum (\text{Bond Enthalpy of bonds broken}) - \sum (\text{Bond Enthalpy of bonds formed})$.
Step2: Identify bonds broken and formed
In the reaction $\ce{H_{2}(g) + Cl_{2}(g) -> 2HCl(g)}$, we break 1 $\ce{H - H}$ bond and 1 $\ce{Cl - Cl}$ bond. We form 2 $\ce{H - Cl}$ bonds.
Step3: Calculate energy required to break bonds
Energy to break 1 $\ce{H - H}$ bond: $436\ \text{kJ/mol}$.
Energy to break 1 $\ce{Cl - Cl}$ bond: $243\ \text{kJ/mol}$.
Total energy for breaking bonds: $436 + 243 = 679\ \text{kJ/mol}$.
Step4: Calculate energy released when forming bonds
Energy released to form 1 $\ce{H - Cl}$ bond: $432\ \text{kJ/mol}$.
For 2 $\ce{H - Cl}$ bonds: $2 \times 432 = 864\ \text{kJ/mol}$.
Step5: Calculate enthalpy change
$\Delta H = \text{Energy for breaking bonds} - \text{Energy for forming bonds} = 679 - 864 = -185\ \text{kJ/mol}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. $-185\ \text{kJ/mol}_{\text{rxn}}$