QUESTION IMAGE
Question
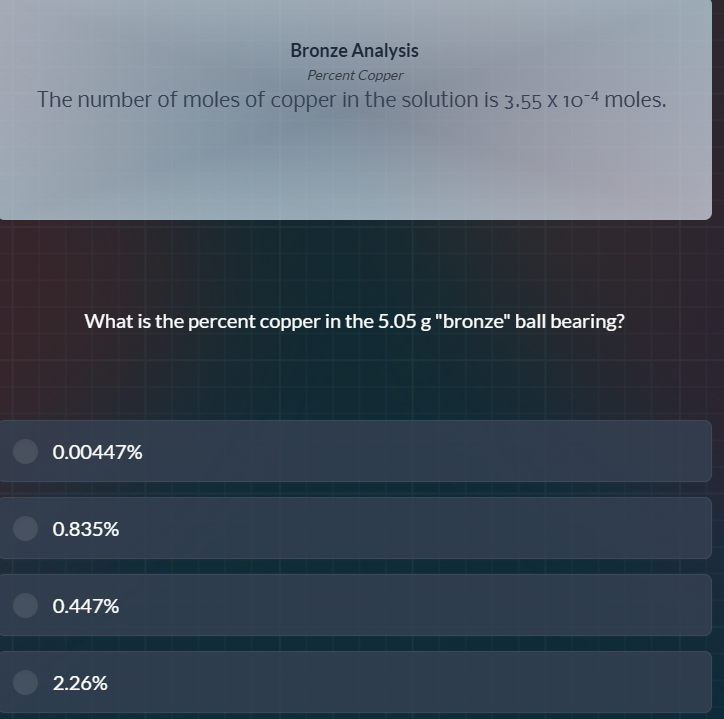
bronze analysis
percent copper
the number of moles of copper in the solution is $3.55 \times 10^{-4}$ moles.
what is the percent copper in the 5.05 g \bronze\ ball bearing?
0.00447%
0.835%
0.447%
2.26%
Step1: Find mass of copper
Molar mass of Cu = 63.55 g/mol.
Mass = moles × molar mass
$\text{Mass of Cu} = 3.55 \times 10^{-4}\ \text{mol} \times 63.55\ \text{g/mol}$
$\text{Mass of Cu} = 0.02256\ \text{g}$
Step2: Calculate percent copper
Percent copper = $\frac{\text{Mass of Cu}}{\text{Mass of bronze}} \times 100\%$
$\text{Percent Cu} = \frac{0.02256\ \text{g}}{5.05\ \text{g}} \times 100\%$
$\text{Percent Cu} \approx 0.447\%$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
0.447%