QUESTION IMAGE
Question
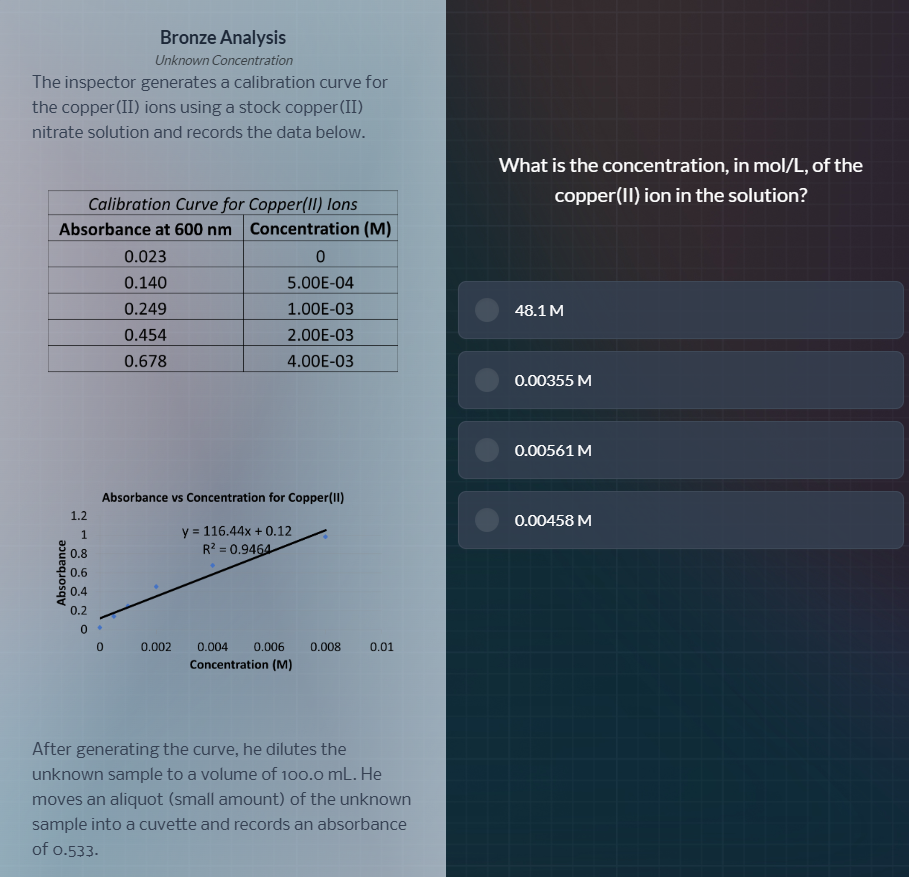
bronze analysis
unknown concentration
the inspector generates a calibration curve for
the copper(ii) ions using a stock copper(ii)
nitrate solution and records the data below.
calibration curve for copper(ii) ions
absorbance at 600 nm | concentration (m)
0.023 | 0
0.140 | 5.00e-04
0.249 | 1.00e-03
0.454 | 2.00e-03
0.678 | 4.00e-03
absorbance vs concentration for copper(ii)
y = 116.44x + 0.12
r² = 0.9464
after generating the curve, he dilutes the
unknown sample to a volume of 100.0 ml. he
moves an aliquot (small amount) of the unknown
sample into a cuvette and records an absorbance
of 0.533.
what is the concentration, in mol/l, of the
copper(ii) ion in the solution?
48.1 m
0.00355 m
0.00561 m
0.00458 m
Step1: Identify calibration equation
The calibration curve equation is $y = 116.44x + 0.12$, where $y$ = absorbance, $x$ = concentration (M).
Step2: Substitute unknown absorbance
Substitute $y = 0.533$ into the equation:
$0.533 = 116.44x + 0.12$
Step3: Isolate the concentration term
Rearrange to solve for $x$:
$116.44x = 0.533 - 0.12$
$116.44x = 0.413$
Step4: Calculate concentration
Solve for $x$:
$x = \frac{0.413}{116.44}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
0.00355 M