QUESTION IMAGE
Question
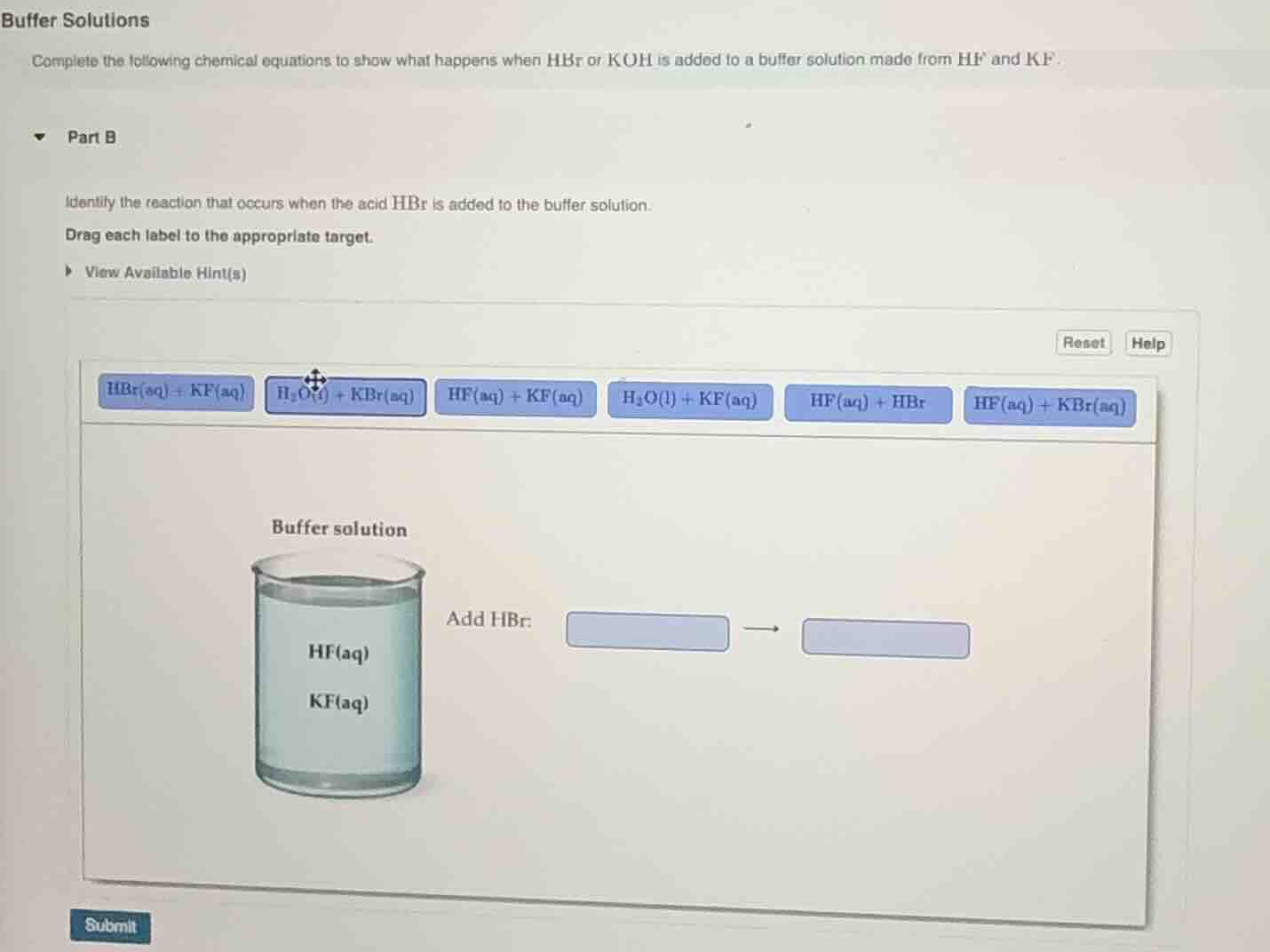
buffer solutions
complete the following chemical equations to show what happens when hbr or koh is added to a buffer solution made from hf and kf.
part b
identify the reaction that occurs when the acid hbr is added to the buffer solution.
drag each label to the appropriate target.
view available hint(s)
reset help
hbr(aq) + kf(aq) h₂o(l) + kbr(aq) hf(aq) + kf(aq) h₂o(l) + kf(aq) hf(aq) + hbr hf(aq) + kbr(aq)
buffer solution
add hbr:
hf(aq)
kf(aq)
submit
Step1: Identify the buffer components
The buffer solution is made of \( \text{HF}(aq) \) (weak acid) and \( \text{KF}(aq) \) (conjugate base, since \( \text{KF} \) dissociates into \( \text{K}^+ \) and \( \text{F}^- \)).
Step2: Analyze the added acid (HBr)
\( \text{HBr} \) is a strong acid, so it will dissociate into \( \text{H}^+ \) and \( \text{Br}^- \) ions. The \( \text{H}^+ \) from \( \text{HBr} \) will react with the conjugate base of the buffer (\( \text{F}^- \) from \( \text{KF} \)).
Step3: Write the reaction
The \( \text{F}^- \) (from \( \text{KF} \)) reacts with \( \text{H}^+ \) (from \( \text{HBr} \)) to form \( \text{HF} \). The \( \text{K}^+ \) and \( \text{Br}^- \) will be spectator ions and form \( \text{KBr} \). So the reaction is:
\( \text{HBr}(aq) + \text{KF}(aq)
ightarrow \text{HF}(aq) + \text{KBr}(aq) \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
First box: \( \text{HBr}(aq) + \text{KF}(aq) \)
Second box: \( \text{HF}(aq) + \text{KBr}(aq) \)