QUESTION IMAGE
Question
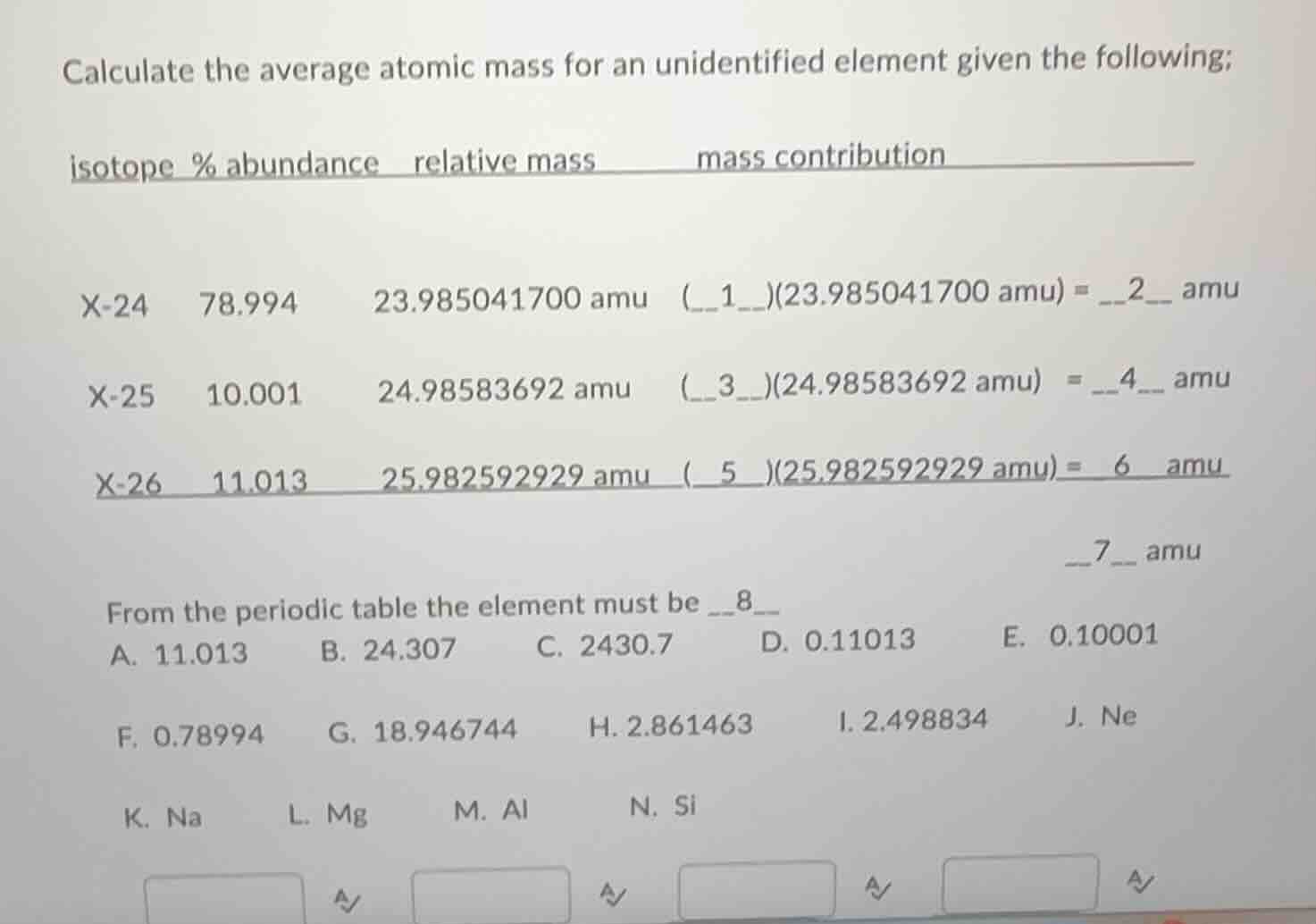
calculate the average atomic mass for an unidentified element given the following;
isotope % abundance relative mass mass contribution
x-24 78.994 23.985041700 amu (1)(23.985041700 amu) = 2 amu
x-25 10.001 24.98583692 amu (3)(24.98583692 amu) = 4 amu
x-26 11.013 25.982592929 amu (5)(25.982592929 amu) = 6 amu
7 amu
from the periodic table the element must be 8
a. 11.013 b. 24.307 c. 2430.7 d. 0.11013 e. 0.10001
f. 0.78994 g. 18.946744 h. 2.861463 i. 2.498834 j. ne
k. na l. mg m. al n. si
Step1: Convert % to decimal for X-24
To find the mass contribution, we first convert the percentage abundance to a decimal by dividing by 100. For X-24, the % abundance is 78.994, so the decimal is $ \frac{78.994}{100} = 0.78994 $.
Step2: Calculate mass contribution for X-24
Multiply the decimal abundance by the relative mass. So, $ 0.78994 \times 23.985041700 \approx 18.946744 $ amu.
Step3: Convert % to decimal for X-25
The % abundance is 10.001, so the decimal is $ \frac{10.001}{100} = 0.10001 $.
Step4: Calculate mass contribution for X-25
Multiply the decimal abundance by the relative mass. So, $ 0.10001 \times 24.98583692 \approx 2.498834 $ amu.
Step5: Convert % to decimal for X-26
The % abundance is 11.013, so the decimal is $ \frac{11.013}{100} = 0.11013 $.
Step6: Calculate mass contribution for X-26
Multiply the decimal abundance by the relative mass. So, $ 0.11013 \times 25.982592929 \approx 2.861463 $ amu.
Step7: Sum the mass contributions
Add the three mass contributions: $ 18.946744 + 2.498834 + 2.861463 \approx 24.307 $ amu.
Step8: Identify the element
Looking at the periodic table, the element with an average atomic mass of approximately 24.307 amu is Magnesium (Mg).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s (for each blank):
- \( 0.78994 \) (Option F)
- \( 18.946744 \) (Option G)
- \( 0.10001 \) (Option E)
- \( 2.498834 \) (Option I)
- \( 0.11013 \) (Option D)
- \( 2.861463 \) (Option H)
- \( 24.307 \) (Option B)
- \( \text{Mg} \) (Option L)