QUESTION IMAGE
Question
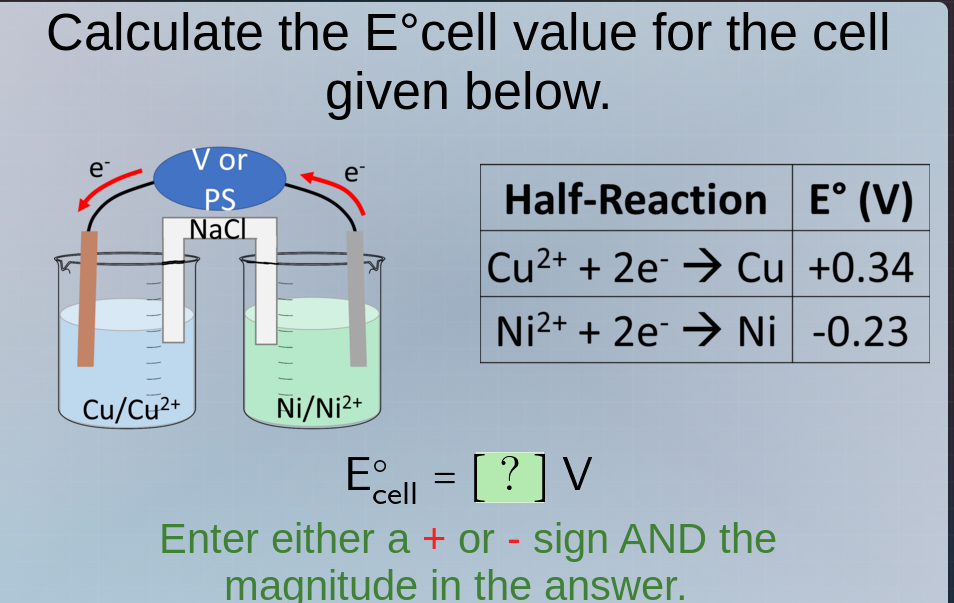
calculate the e°cell value for the cell given below. half-reaction e° (v) cu²⁺ + 2e⁻ → cu +0.34 ni²⁺ + 2e⁻ → ni -0.23 e°cell = ? v enter either a + or - sign and the magnitude in the answer.
Step1: Identify anode and cathode
In a galvanic cell, the more positive reduction potential is for the cathode (reduction), and the less positive (or more negative) is for the anode (oxidation). Here, \( \text{Cu}^{2+} + 2e^-
ightarrow \text{Cu} \) has \( E^\circ = +0.34 \, \text{V} \) (cathode, reduction), and \( \text{Ni}
ightarrow \text{Ni}^{2+} + 2e^- \) (anode, oxidation, reverse of \( \text{Ni}^{2+} + 2e^-
ightarrow \text{Ni} \) with \( E^\circ_{\text{oxidation}} = +0.23 \, \text{V} \)).
Step2: Calculate \( E^\circ_{\text{cell}} \)
The formula for \( E^\circ_{\text{cell}} \) is \( E^\circ_{\text{cathode (reduction)}} - E^\circ_{\text{anode (reduction)}} \) (or \( E^\circ_{\text{cathode}} + E^\circ_{\text{anode (oxidation)}} \)). Using \( E^\circ_{\text{cell}} = E^\circ_{\text{cathode (reduction)}} - E^\circ_{\text{anode (reduction)}} \):
\( E^\circ_{\text{cell}} = 0.34 - (-0.23) \)
\( E^\circ_{\text{cell}} = 0.34 + 0.23 \)
\( E^\circ_{\text{cell}} = 0.57 \, \text{V} \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( +0.57 \)