QUESTION IMAGE
Question
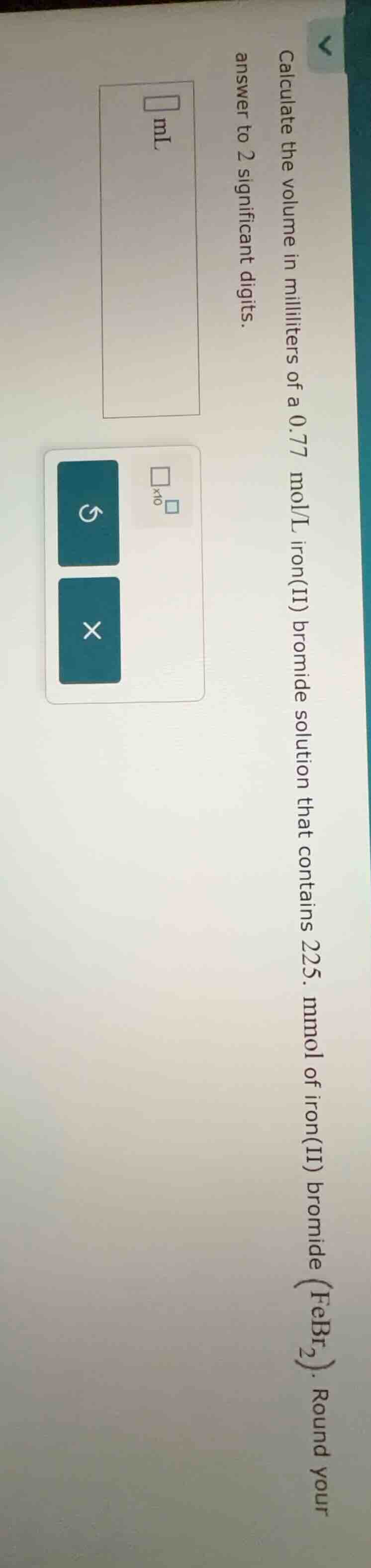
calculate the volume in milliliters of a 0.77 mol/l iron(ii) bromide solution that contains 225. mmol of iron(ii) bromide ($febr_2$). round your answer to 2 significant digits.
Step1: Convert mmol to mol
$225. \text{ mmol} = 225. \times 10^{-3} \text{ mol} = 0.225 \text{ mol}$
Step2: Use molarity formula to find volume
Molarity $M = \frac{n}{V}$, rearrange to $V = \frac{n}{M}$
$V = \frac{0.225 \text{ mol}}{0.77 \text{ mol/L}} \approx 0.2922 \text{ L}$
Step3: Convert L to mL
$0.2922 \text{ L} \times 1000 = 292.2 \text{ mL}$
Step4: Round to 2 significant digits
$292.2 \text{ mL} \approx 290 \text{ mL}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
290 mL