QUESTION IMAGE
Question
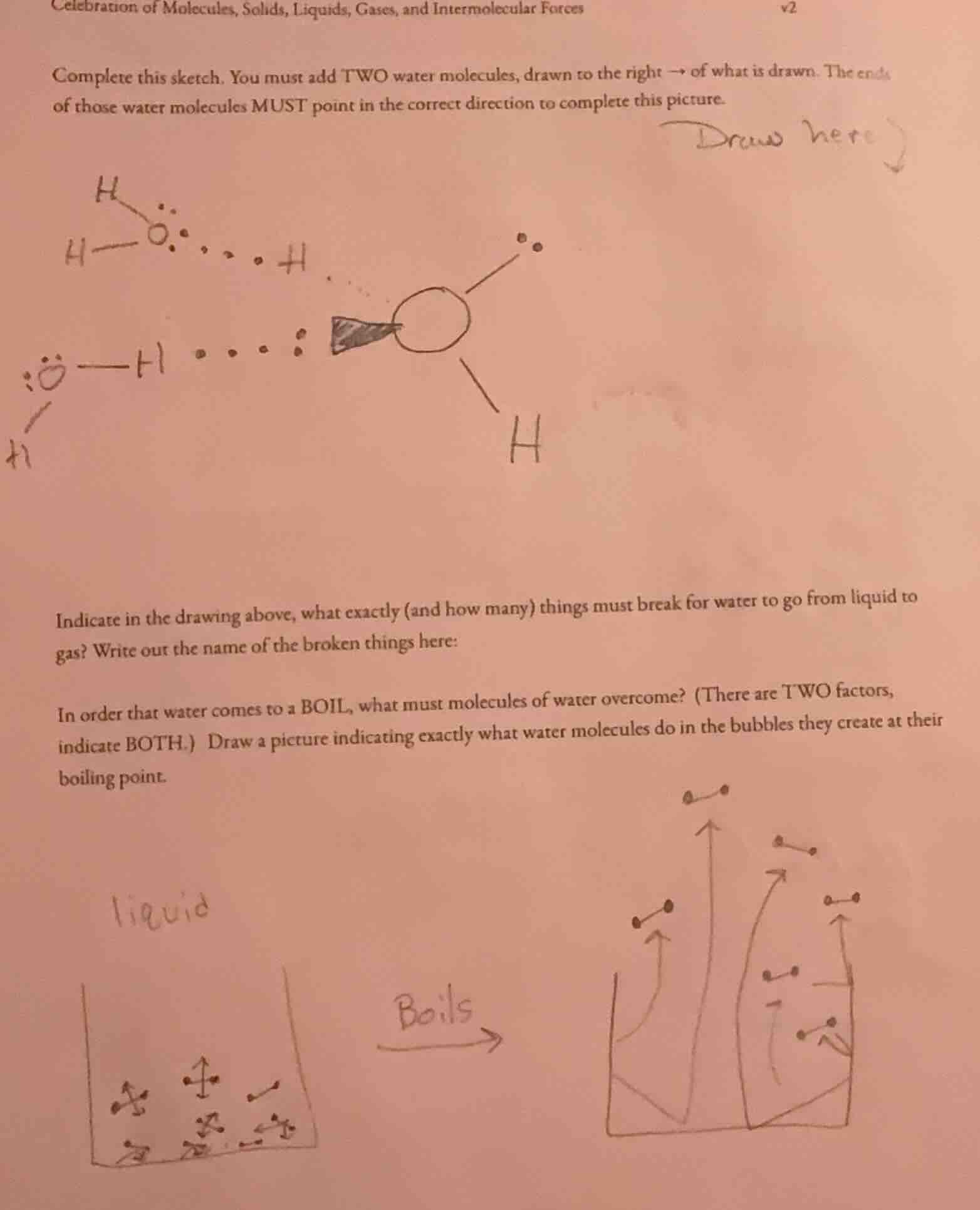
celebration of molecules, solids, liquids, gases, and intermolecular forces v2
complete this sketch. you must add two water molecules, drawn to the right → of what is drawn. the ends of those water molecules must point in the correct direction to complete this picture.
indicate in the drawing above, what exactly (and how many) things must break for water to go from liquid to gas? write out the name of the broken things here:
in order that water comes to a boil, what must molecules of water overcome? (there are two factors, indicate both.) draw a picture indicating exactly what water molecules do in the bubbles they create at their boiling point.
Part 1: Adding Water Molecules (Intermolecular Forces - Hydrogen Bonding)
Water molecules (\(H_2O\)) have a bent structure with a partial negative charge on the oxygen (\(O\)) and partial positive charges on the hydrogens (\(H\)). Hydrogen bonds form between the partially positive \(H\) of one water molecule and the partially negative \(O\) of another.
To draw the two water molecules to the right:
- For the first new water molecule: The \(H\) atoms should point toward the lone pairs of the existing \(O\) (to form hydrogen bonds), and the \(O\) should have two lone pairs.
- For the second new water molecule: Similarly, align the \(H\) atoms to form hydrogen bonds with the previous water molecule’s \(O\) or lone pairs, ensuring the dipole direction (partial + on \(H\), partial - on \(O\)) is correct.
Part 2: Breaking for Liquid to Gas (Vaporization)
When water goes from liquid to gas (vaporization), the intermolecular forces (specifically hydrogen bonds) between water molecules must break. In the drawing, each dashed line (hydrogen bond) between water molecules is what breaks. If there are, say, \(n\) hydrogen bonds between the drawn molecules, that’s how many break (but the key is identifying the force: hydrogen bonds).
Part 3: Boiling Factors and Bubble Drawing
To boil, water molecules must overcome:
- Intermolecular forces (hydrogen bonds) (between molecules).
- Atmospheric pressure (the external pressure pushing down on the liquid).
For the bubble drawing: In the liquid, molecules are close with hydrogen bonds. At boiling, bubbles form when groups of water molecules (in the gas phase, \(H_2O\) vapor) escape. Draw a bubble (a circle) with \(H_2O\) molecules inside, spaced farther apart than in the liquid, with arrows showing them escaping (vaporizing) while breaking hydrogen bonds.
Final Answers (Key Concepts)
- Broken for liquid to gas: Hydrogen bonds (intermolecular forces).
- Boiling factors: Intermolecular forces (hydrogen bonds) and atmospheric pressure.
- Bubble drawing: Show liquid water with close molecules (hydrogen bonds), a bubble with gas-phase \(H_2O\) molecules (farther apart) escaping, breaking hydrogen bonds.
(Note: For the drawing, visualize or sketch the water molecules with correct hydrogen bonding orientation, label hydrogen bonds, and show vaporization in bubbles.)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Part 1: Adding Water Molecules (Intermolecular Forces - Hydrogen Bonding)
Water molecules (\(H_2O\)) have a bent structure with a partial negative charge on the oxygen (\(O\)) and partial positive charges on the hydrogens (\(H\)). Hydrogen bonds form between the partially positive \(H\) of one water molecule and the partially negative \(O\) of another.
To draw the two water molecules to the right:
- For the first new water molecule: The \(H\) atoms should point toward the lone pairs of the existing \(O\) (to form hydrogen bonds), and the \(O\) should have two lone pairs.
- For the second new water molecule: Similarly, align the \(H\) atoms to form hydrogen bonds with the previous water molecule’s \(O\) or lone pairs, ensuring the dipole direction (partial + on \(H\), partial - on \(O\)) is correct.
Part 2: Breaking for Liquid to Gas (Vaporization)
When water goes from liquid to gas (vaporization), the intermolecular forces (specifically hydrogen bonds) between water molecules must break. In the drawing, each dashed line (hydrogen bond) between water molecules is what breaks. If there are, say, \(n\) hydrogen bonds between the drawn molecules, that’s how many break (but the key is identifying the force: hydrogen bonds).
Part 3: Boiling Factors and Bubble Drawing
To boil, water molecules must overcome:
- Intermolecular forces (hydrogen bonds) (between molecules).
- Atmospheric pressure (the external pressure pushing down on the liquid).
For the bubble drawing: In the liquid, molecules are close with hydrogen bonds. At boiling, bubbles form when groups of water molecules (in the gas phase, \(H_2O\) vapor) escape. Draw a bubble (a circle) with \(H_2O\) molecules inside, spaced farther apart than in the liquid, with arrows showing them escaping (vaporizing) while breaking hydrogen bonds.
Final Answers (Key Concepts)
- Broken for liquid to gas: Hydrogen bonds (intermolecular forces).
- Boiling factors: Intermolecular forces (hydrogen bonds) and atmospheric pressure.
- Bubble drawing: Show liquid water with close molecules (hydrogen bonds), a bubble with gas-phase \(H_2O\) molecules (farther apart) escaping, breaking hydrogen bonds.
(Note: For the drawing, visualize or sketch the water molecules with correct hydrogen bonding orientation, label hydrogen bonds, and show vaporization in bubbles.)