QUESTION IMAGE
Question
chapter 4 and 9 homework
exercise 4.52 - enhanced - with feedback
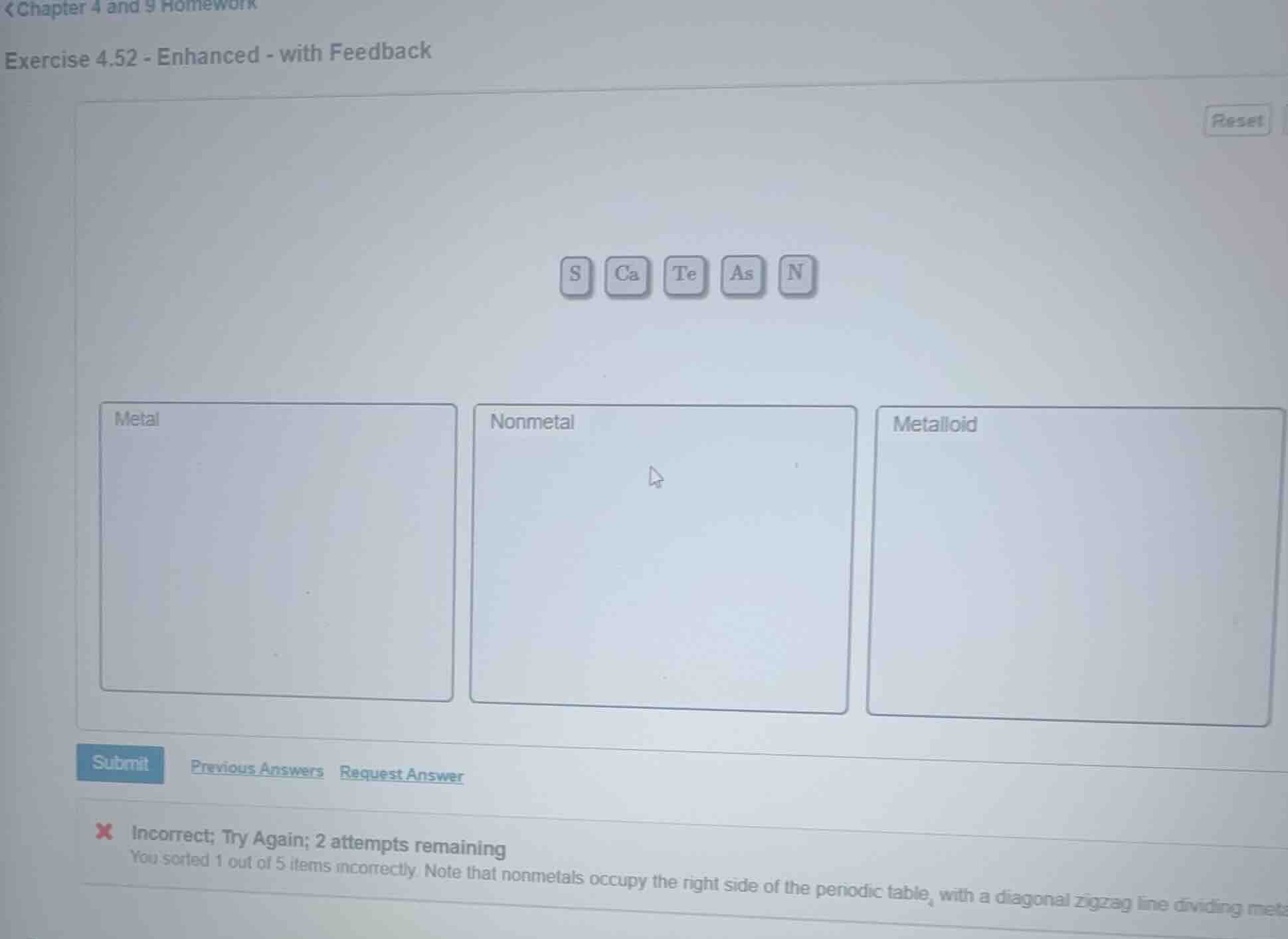
s ca te as n
metal nonmetal metalloid
submit previous answers request answer
incorrect; try again; 2 attempts remaining
you sorted 1 out of 5 items incorrectly. note that nonmetals occupy the right side of the periodic table, with a diagonal zigzag line dividing meta
Brief Explanations
Classify each element based on periodic table group/position:
- Ca is an alkaline earth metal (Group 2).
- S, N are nonmetals (right side of periodic table, no metallic properties).
- As is a metalloid (lies on the zigzag dividing line).
- Te is a metalloid (lies on the zigzag dividing line).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Metal: Ca
Nonmetal: S, N
Metalloid: As, Te