QUESTION IMAGE
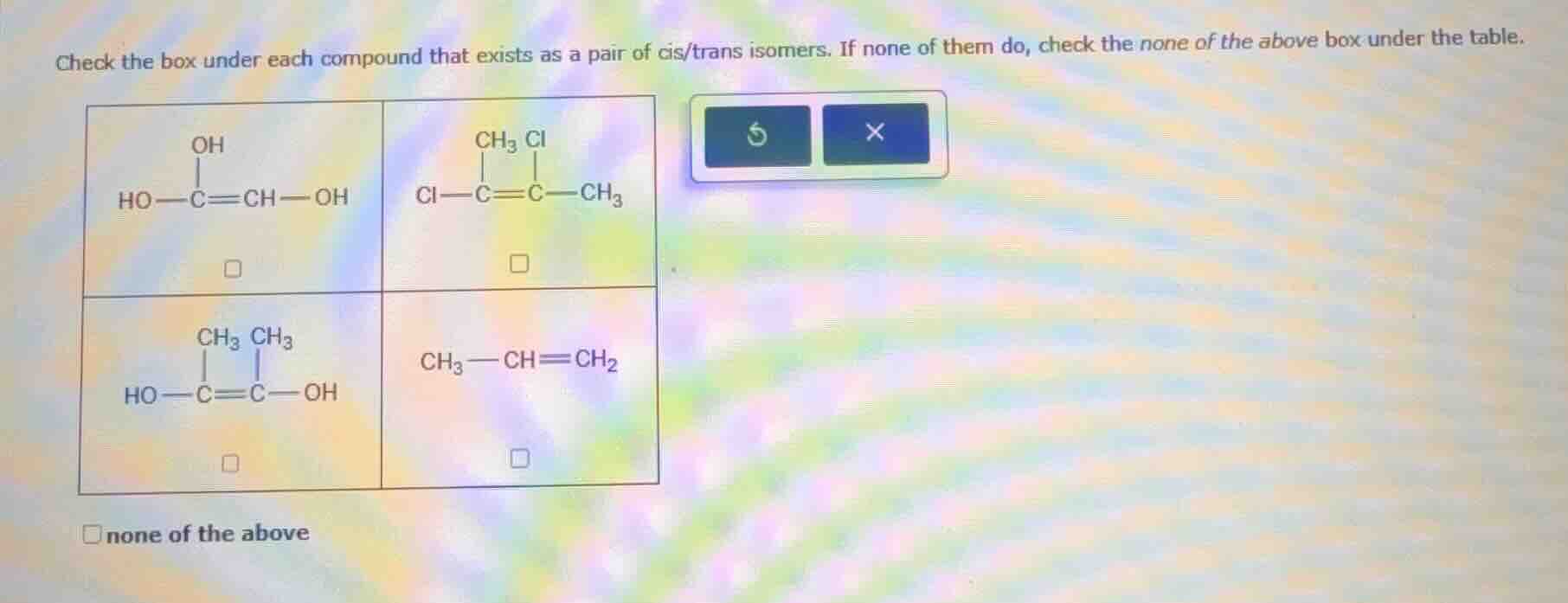
Question
check the box under each compound that exists as a pair of cis/trans isomers. if none of them do, check the none of the above box under the table.
ho−c(oh)=ch−oh
□
cl−c(ch₃, cl)=c−ch₃
□
ho−c(ch₃, ch₃)=c−oh
□
ch₃−ch=ch₂
□
□none of the above
To determine if a compound has cis/trans isomers, we check the following conditions for the double bond (\(C = C\)):
- Each carbon in the double bond must be bonded to two different groups (atoms or groups of atoms).
Step 1: Analyze the first compound (\(HO - C = CH - OH\))
The left carbon in \(C = C\) is bonded to \(HO -\) and \(OH\), and the right carbon is bonded to \(H\) and \( - OH\). Wait, no, let's re - examine. The left carbon: \(HO - C=\) (so bonded to \(HO -\) and \(O\) (from \(OH\) on itself? Wait, no, the structure is \(HO - C(OH)=CH - OH\). Wait, the double - bonded carbons: the first \(C\) (with \(OH\) and \(HO -\)) and the second \(C\) (with \(H\) and \( - OH\)). The second carbon is bonded to \(H\) and \( - OH\), and the first is bonded to \(HO -\) and \(OH\). But the key is: for cis/trans, each carbon of the double bond must have two different substituents. Wait, the second carbon (\(CH\)) is bonded to \(H\) and \( - CH - OH\)? No, maybe I misread. Let's look at the second compound: \(Cl - C(CH_3)=C(Cl)-CH_3\). Wait, no, the second compound is \(Cl - C(CH_3)=C(Cl)-CH_3\)? Wait, the structure is \(Cl - C(CH_3)=C(Cl)-CH_3\)? No, the given structure is \(Cl - C(CH_3)=C(Cl)-CH_3\)? Wait, no, the second compound is \(Cl - C(CH_3)=C(Cl)-CH_3\)? Wait, the first compound: \(HO - C(OH)=CH - OH\). The double - bonded carbons: \(C(OH)\) and \(CH\). The \(CH\) carbon is bonded to \(H\) and \( - CH - OH\)? No, maybe a better approach:
For a compound to have cis - trans isomerism, the two carbons of the double bond must each have two different groups attached.
- First compound: \(HO - C(OH)=CH - OH\)
- The carbon on the left of the double bond (\(C\)): bonded to \(HO -\) and \(OH\) (two different groups? Wait, \(HO -\) and \(OH\) are similar in a way, but more importantly, the carbon on the right (\(CH\)) is bonded to \(H\) and \( - CH - OH\)? No, actually, the right - hand carbon of the double bond is bonded to \(H\) and \( - OH\) (from the \(CH - OH\) group). Wait, no, the structure is \(HO - C(OH)=CH - OH\), so the double - bonded carbons: \(C_1\) (with \(HO -\) and \(OH\)) and \(C_2\) (with \(H\) and \( - OH\)). The \(C_2\) is bonded to \(H\) and \( - OH\), and \(C_1\) is bonded to \(HO -\) and \(OH\). But the key is: for \(C = C\) isomerism, each carbon must have two different substituents. The \(C_2\) has \(H\) and \( - OH\) (different), and \(C_1\) has \(HO -\) and \(OH\) (different). Wait, but maybe I made a mistake. Let's check the second compound: \(Cl - C(CH_3)=C(Cl)-CH_3\) (wait, no, the structure is \(Cl - C(CH_3)=C(Cl)-CH_3\)? No, the given structure is \(Cl - C(CH_3)=C(Cl)-CH_3\)? Wait, the second compound is \(Cl - C(CH_3)=C(Cl)-CH_3\)? Wait, the correct way: the second compound is \(Cl - C(CH_3)=C(Cl)-CH_3\)? No, the structure is \(Cl - C(CH_3)=C(Cl)-CH_3\)? Wait, the double - bonded carbons: each carbon is bonded to two different groups? The left carbon: \(Cl\) and \(CH_3\), the right carbon: \(Cl\) and \(CH_3\). Wait, no, that would be the same groups on each carbon? Wait, no, the left carbon: \(Cl\) and \(CH_3\), the right carbon: \(Cl\) and \(CH_3\). So it is a case of cis - trans isomerism because the two carbons of the double bond have the same two substituents (\(Cl\) and \(CH_3\)) but in different arrangements. Wait, no, if both carbons have the same two substituents, then cis - trans is possible. Wait, let's check the third compound: \(HO - C(CH_3)=C(CH_3)-OH\). The two carbons of the double bond are each bonded to \(CH_3\) and \(HO -\) (for the first \(C\)) and \(CH_3\) and \( - OH\) (for the…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The box under the compound \(Cl - C(CH_3)=C(Cl)-CH_3\) (the second compound in the table) should be checked.