QUESTION IMAGE
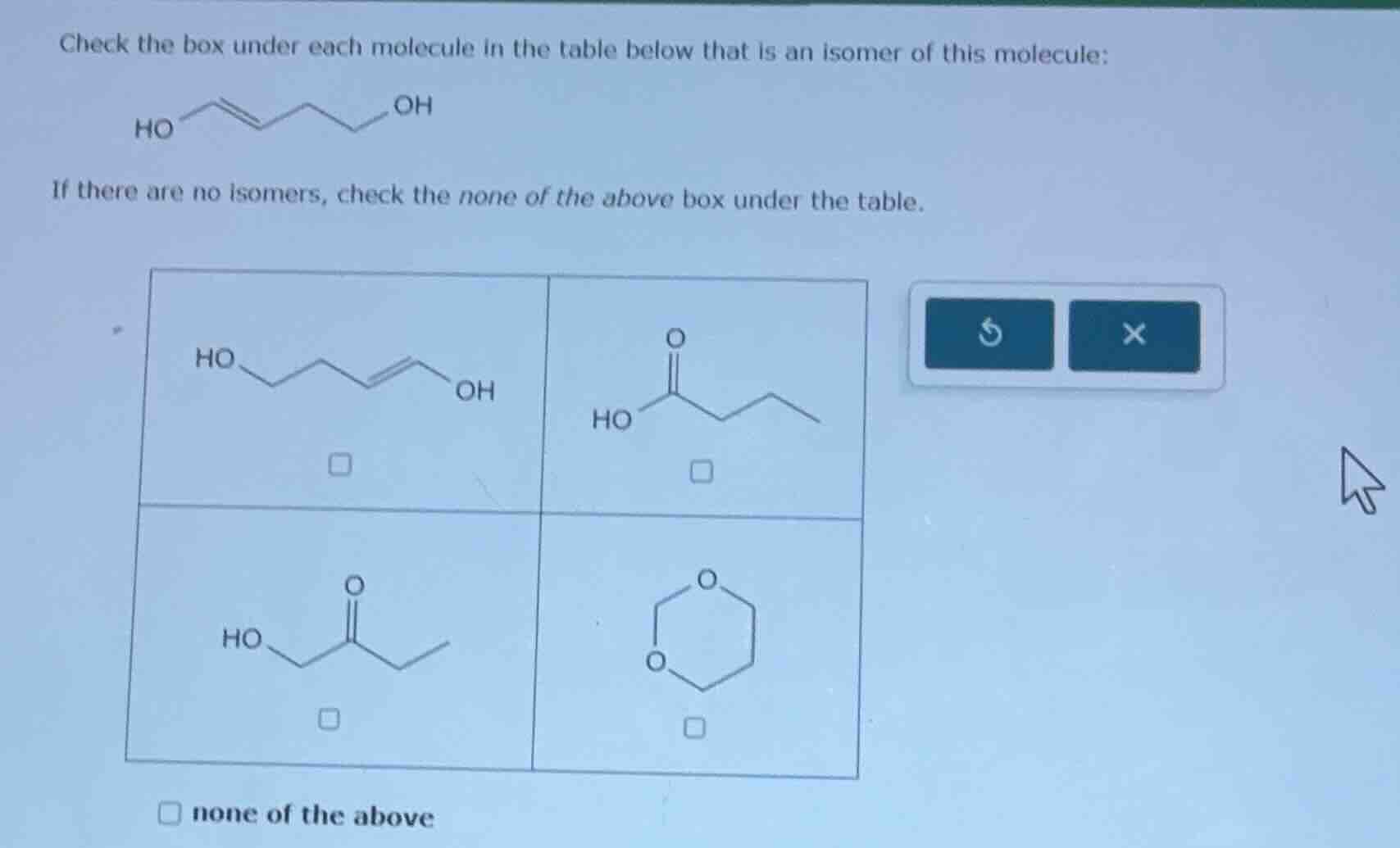
Question
check the box under each molecule in the table below that is an isomer of this molecule:
ho
oh
if there are no isomers, check the none of the above box under the table.
ho
oh
ho
ho
none of the above
To determine isomers, we first find the molecular formula of the given molecule. The given molecule has a double bond, two -OH groups, and a carbon chain. Let's analyze each candidate:
Step 1: Analyze the first candidate (top - left)
The first molecule (top - left) has the same functional groups (two -OH, a double bond) and the same number of carbon, hydrogen, and oxygen atoms as the given molecule. It is a geometric isomer (cis - trans isomer) of the given molecule since the double bond's configuration might be different, but the molecular formula is the same.
Step 2: Analyze the second candidate (top - right)
The top - right molecule has a carboxylic acid group (-COOH) instead of a double bond and an -OH. The molecular formula will be different (it has a carbonyl group (C = O) and a single -OH, while the original has two -OH and a double bond). So, it's not an isomer.
Step 3: Analyze the third candidate (bottom - left)
The bottom - left molecule has a ketone group (C = O) and a single -OH. The molecular formula will be different from the original (original has two -OH and a double bond). So, it's not an isomer.
Step 4: Analyze the fourth candidate (bottom - right)
The bottom - right molecule is a cyclic ether. The number of carbon, hydrogen, and oxygen atoms is different from the original (original has an acyclic structure with a double bond and two -OH). So, it's not an isomer.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The box under the top - left molecule (the one with two -OH groups and a double bond, \(\ce{HO - CH2 - CH2 - CH=CH - OH}\) - like structure) should be checked. Also, if we consider the "none of the above" is not correct here as we have an isomer in the top - left.