QUESTION IMAGE
Question
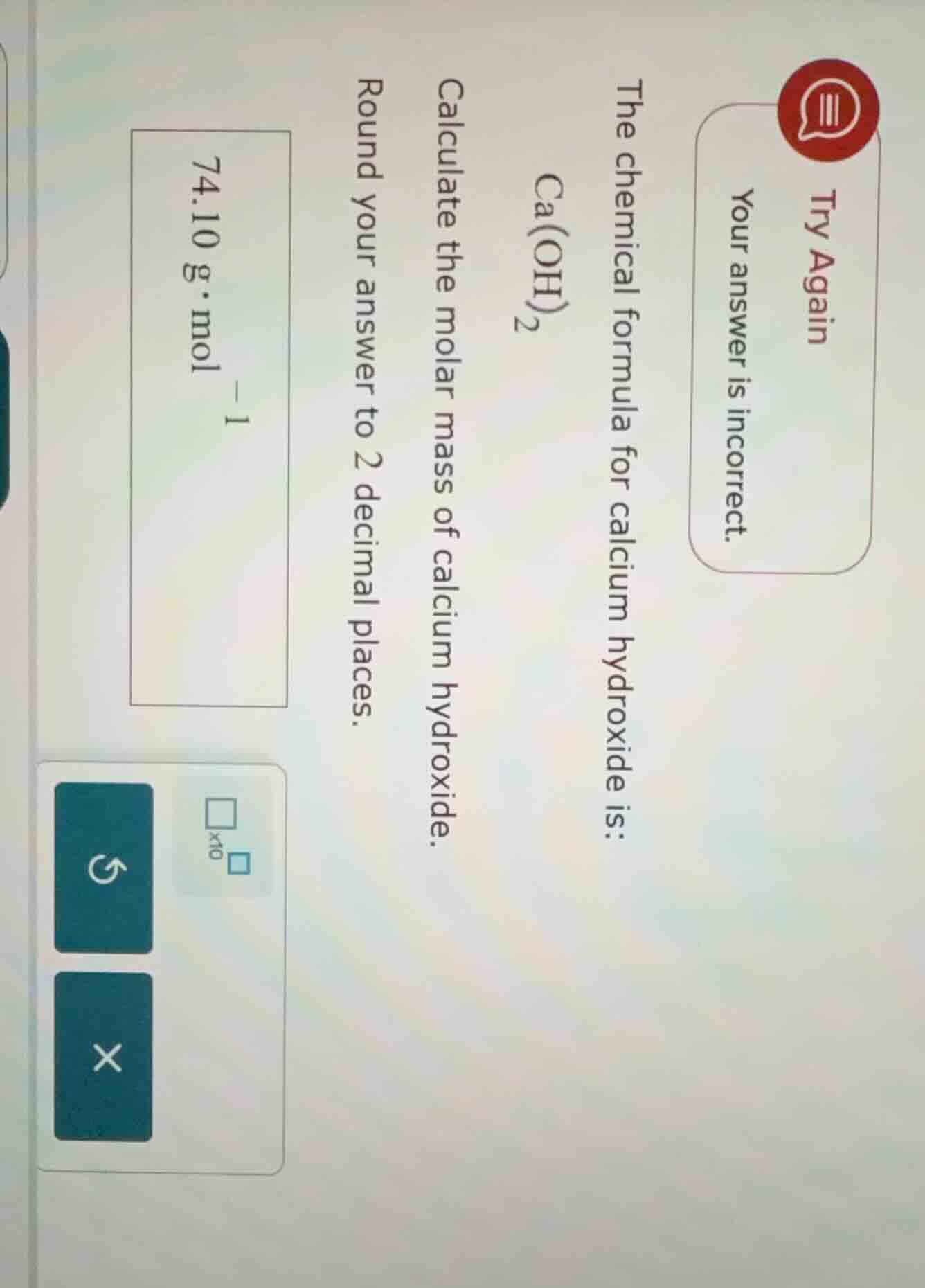
the chemical formula for calcium hydroxide is: ca(oh)₂ calculate the molar mass of calcium hydroxide. round your answer to 2 decimal places. 74.10 g·mol⁻¹
Step1: Identify atomic masses
Atomic mass of Ca: $40.08\ \text{g/mol}$, O: $16.00\ \text{g/mol}$, H: $1.008\ \text{g/mol}$.
Step2: Calculate molar mass of $\ce{Ca(OH)2}$
Molar mass = Mass of Ca + 2×(Mass of O + Mass of H)
$= 40.08 + 2\times(16.00 + 1.008)$
$= 40.08 + 2\times17.008$
$= 40.08 + 34.016$
$= 74.096\ \text{g/mol}$
Step3: Round to 2 decimal places
$74.096$ rounded to 2 decimal places is $74.10\ \text{g/mol}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$74.10\ \text{g/mol}$