QUESTION IMAGE
Question
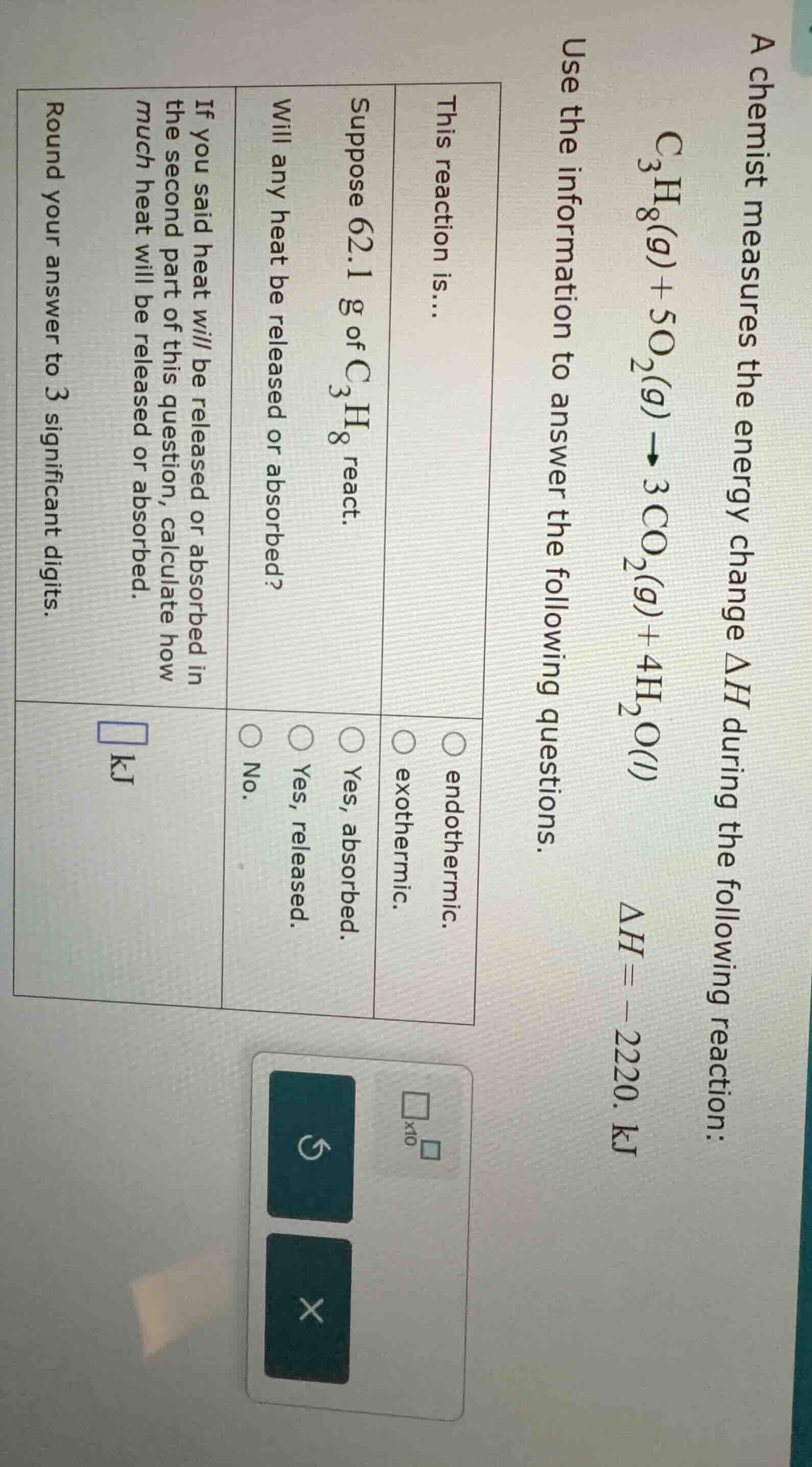
a chemist measures the energy change $\delta h$ during the following reaction: $\ce{c_{3}h_{8}(g) + 5o_{2}(g) \
ightarrow 3co_{2}(g) + 4h_{2}o(l)}$ $\delta h = -2220.\\ \text{kj}$ use the information to answer the following questions. this reaction is... \�LXI0� endothermic. \�LXI1� exothermic. suppose 62.1 g of $\ce{c_{3}h_{8}}$ react. will any heat be released or absorbed? \\(\circ\\) yes, absorbed. \\(\circ\\) yes, released. \\(\circ\\) no. if you said heat will be released or absorbed in the second part of this question, calculate how much heat will be released or absorbed. round your answer to 3 significant digits. \\(\square\\ \text{kj}\\)
Step1: Determine reaction type
The enthalpy change $\Delta H = -2220. \, \text{kJ}$. A negative $\Delta H$ means the reaction is exothermic (heat is released).
Step2: Calculate moles of $\boldsymbol{C_3H_8}$
Molar mass of $C_3H_8$: $3(12.01) + 8(1.008) = 36.03 + 8.064 = 44.094 \, \text{g/mol}$.
Moles of $C_3H_8$: $\frac{62.1 \, \text{g}}{44.094 \, \text{g/mol}} \approx 1.408 \, \text{mol}$.
Step3: Calculate heat released
From the reaction, 1 mol of $C_3H_8$ releases $2220. \, \text{kJ}$.
Heat released: $1.408 \, \text{mol} \times 2220. \, \text{kJ/mol} \approx 3125.76 \, \text{kJ}$.
Round to 3 significant figures: $3130 \, \text{kJ}$ (or $3.13 \times 10^3 \, \text{kJ}$).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Reaction type: exothermic
- Heat release: Yes, released
- Heat amount: $\boldsymbol{3130 \, \text{kJ}}$ (or $\boldsymbol{3.13 \times 10^3 \, \text{kJ}}$)