QUESTION IMAGE
Question
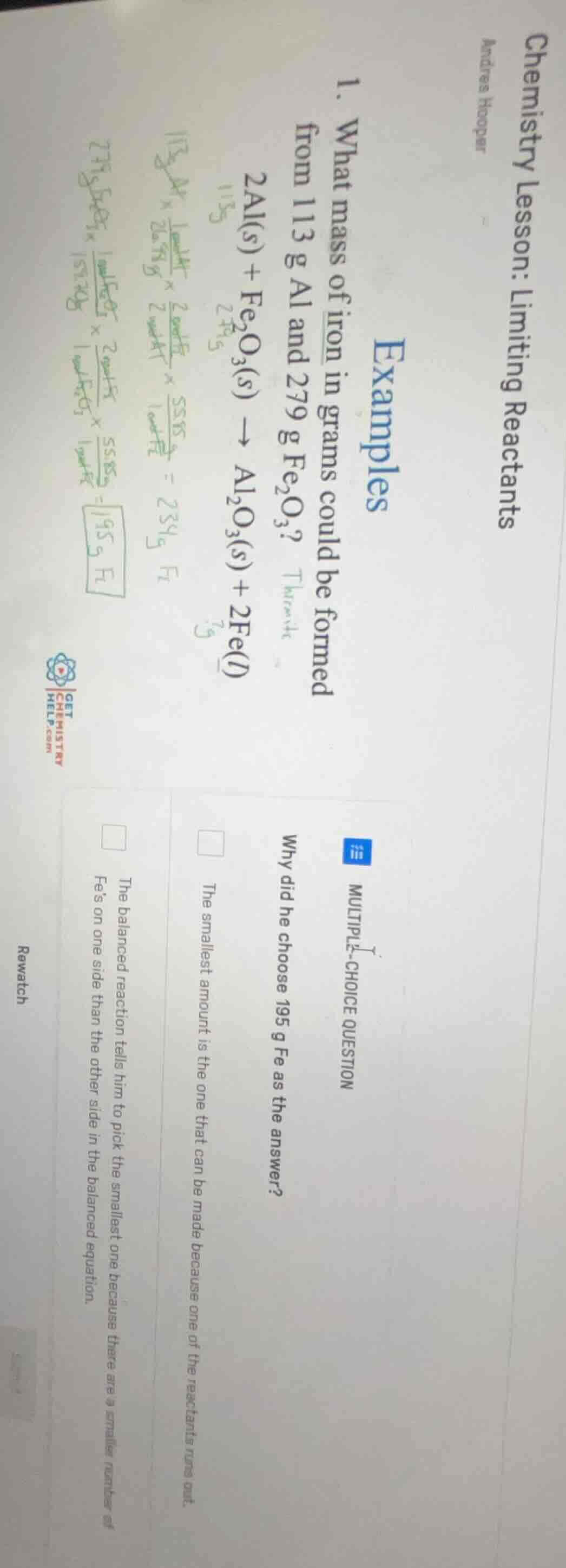
chemistry lesson: limiting reactants
andres hooper
examples
- what mass of iron in grams could be formed from 113 g al and 279 g $fe_2o_3$?
$2al(s) + fe_2o_3(s) \
ightarrow al_2o_3(s) + 2fe(l)$
multiple-choice question
why did he choose 195 g fe as the answer?
□ the smallest amount is the one that can be made because one of the reactants runs out.
□ the balanced reaction tells him to pick the smallest one because there are a smaller number of fes on one side than the other side in the balanced equation.
In limiting reactant problems, the maximum amount of product that can be formed is determined by the reactant that runs out first (the limiting reactant). The smaller calculated mass of product corresponds to the limiting reactant, as this is the maximum quantity that can actually be produced before one reactant is fully consumed.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The smallest amount is the one that can be made because one of the reactants runs out.