QUESTION IMAGE
Question
chemistry lesson: limiting reactants
andres hooper
examples
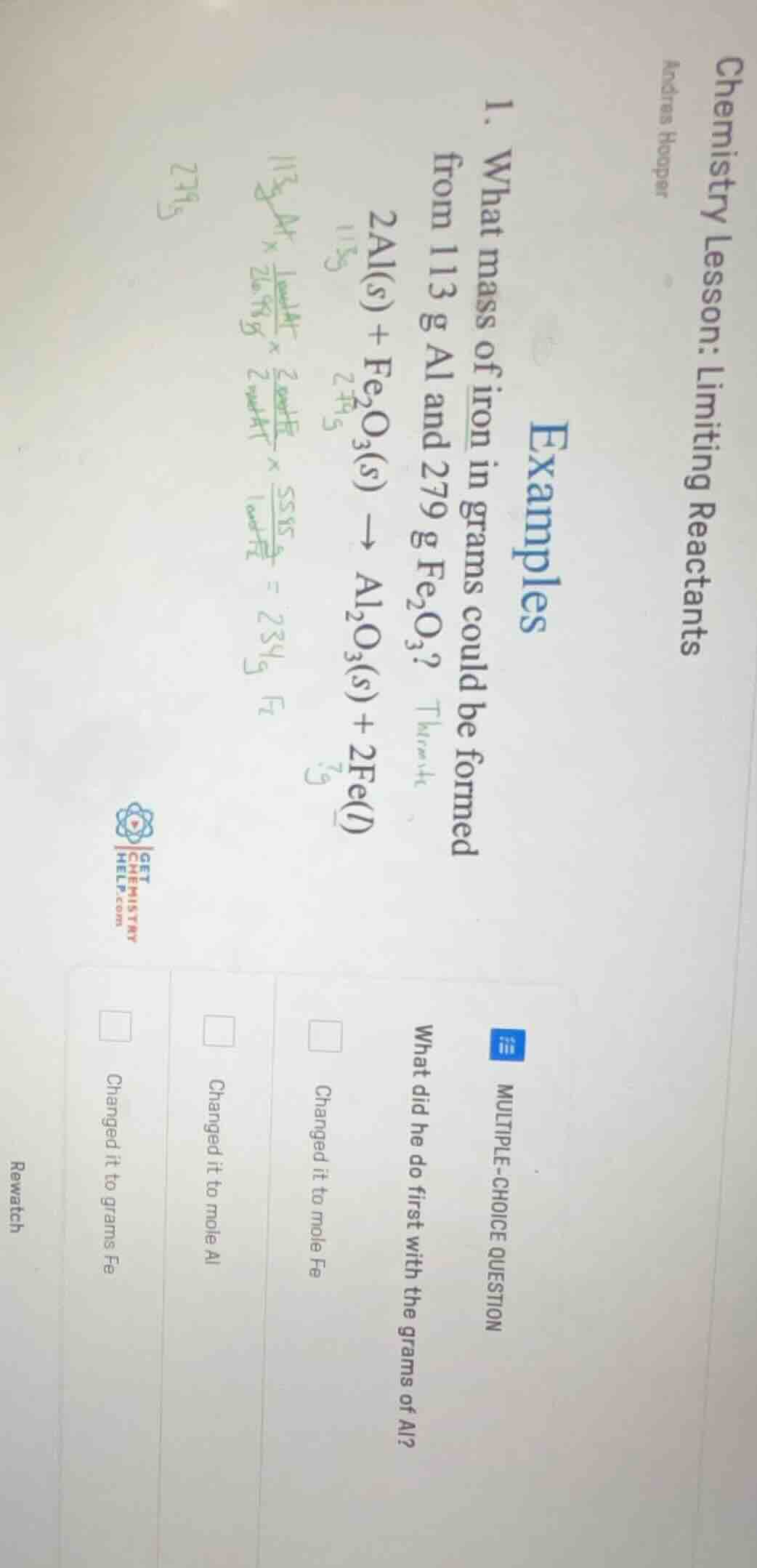
- what mass of iron in grams could be formed from 113 g al and 279 g fe₂o₃?
2al(s) + fe₂o₃(s) → al₂o₃(s) + 2fe(l)
multiple-choice question
what did he do first with the grams of al?
□ changed it to mole fe
□ changed it to mole al
□ changed it to grams fe
To solve limiting reactant problems, the first step is to convert the given mass of a reactant to moles using its molar mass. Looking at the handwritten calculation for Al, the first conversion is $\frac{1\ \text{mol Al}}{26.98\ \text{g Al}}$ applied to 113 g Al, which converts grams of Al to moles of Al.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. Changed it to mole Al