QUESTION IMAGE
Question
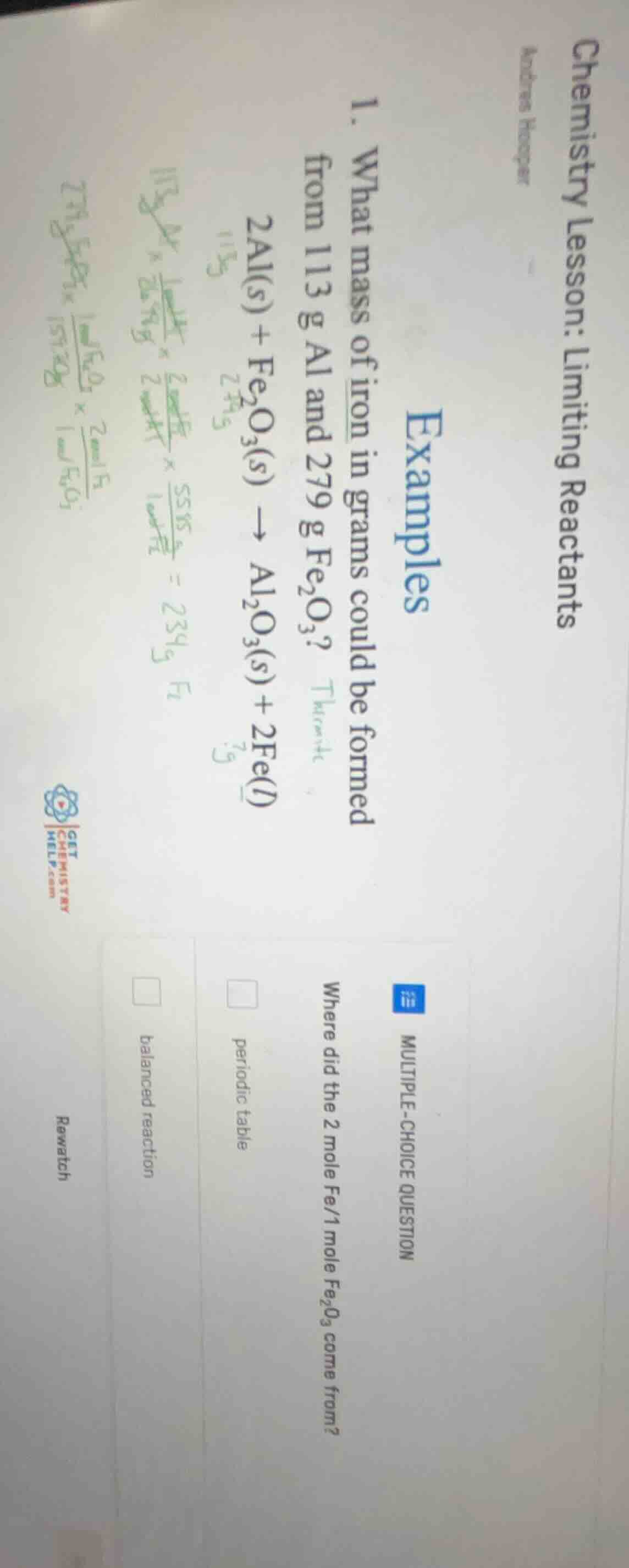
chemistry lesson: limiting reactants
andrew hooper
examples
- what mass of iron in grams could be formed from 113 g al and 279 g $fe_2o_3$?
$2al(s) + fe_2o_3(s) \
ightarrow al_2o_3(s) + 2fe(l)$
multiple-choice question
where did the 2 mole fe/1 mole $fe_2o_3$ come from?
□ periodic table
□ balanced reaction
First, solving the mass calculation problem:
Step1: Calculate moles of Al
Molar mass of Al = 26.98 g/mol
$n_{\text{Al}} = \frac{113\ \text{g}}{26.98\ \text{g/mol}} \approx 4.19\ \text{mol}$
Step2: Calculate moles of $\text{Fe}_2\text{O}_3$
Molar mass of $\text{Fe}_2\text{O}_3 = 2\times55.85 + 3\times16.00 = 159.7\ \text{g/mol}$
$n_{\text{Fe}_2\text{O}_3} = \frac{279\ \text{g}}{159.7\ \text{g/mol}} \approx 1.75\ \text{mol}$
Step3: Find limiting reactant
From balanced equation: $2\text{Al} \sim 1\text{Fe}_2\text{O}_3$
Moles of Al needed for 1.75 mol $\text{Fe}_2\text{O}_3$: $2\times1.75 = 3.50\ \text{mol}$ (we have 4.19 mol, so $\text{Fe}_2\text{O}_3$ is limiting)
Step4: Calculate mass of Fe
From equation: $1\text{Fe}_2\text{O}_3 \sim 2\text{Fe}$
Moles of Fe = $2\times1.75 = 3.50\ \text{mol}$
Molar mass of Fe = 55.85 g/mol
$m_{\text{Fe}} = 3.50\ \text{mol} \times 55.85\ \text{g/mol} = 195.48\ \text{g} \approx 195\ \text{g}$
Then, answering the multiple-choice question:
The mole ratio of $\text{Fe}$ to $\text{Fe}_2\text{O}_3$ is derived from the coefficients of the balanced chemical reaction equation, which shows 1 mole of $\text{Fe}_2\text{O}_3$ produces 2 moles of $\text{Fe}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Mass of iron:
195 g
Multiple-choice answer:
balanced reaction