QUESTION IMAGE
Question
chemistry resources
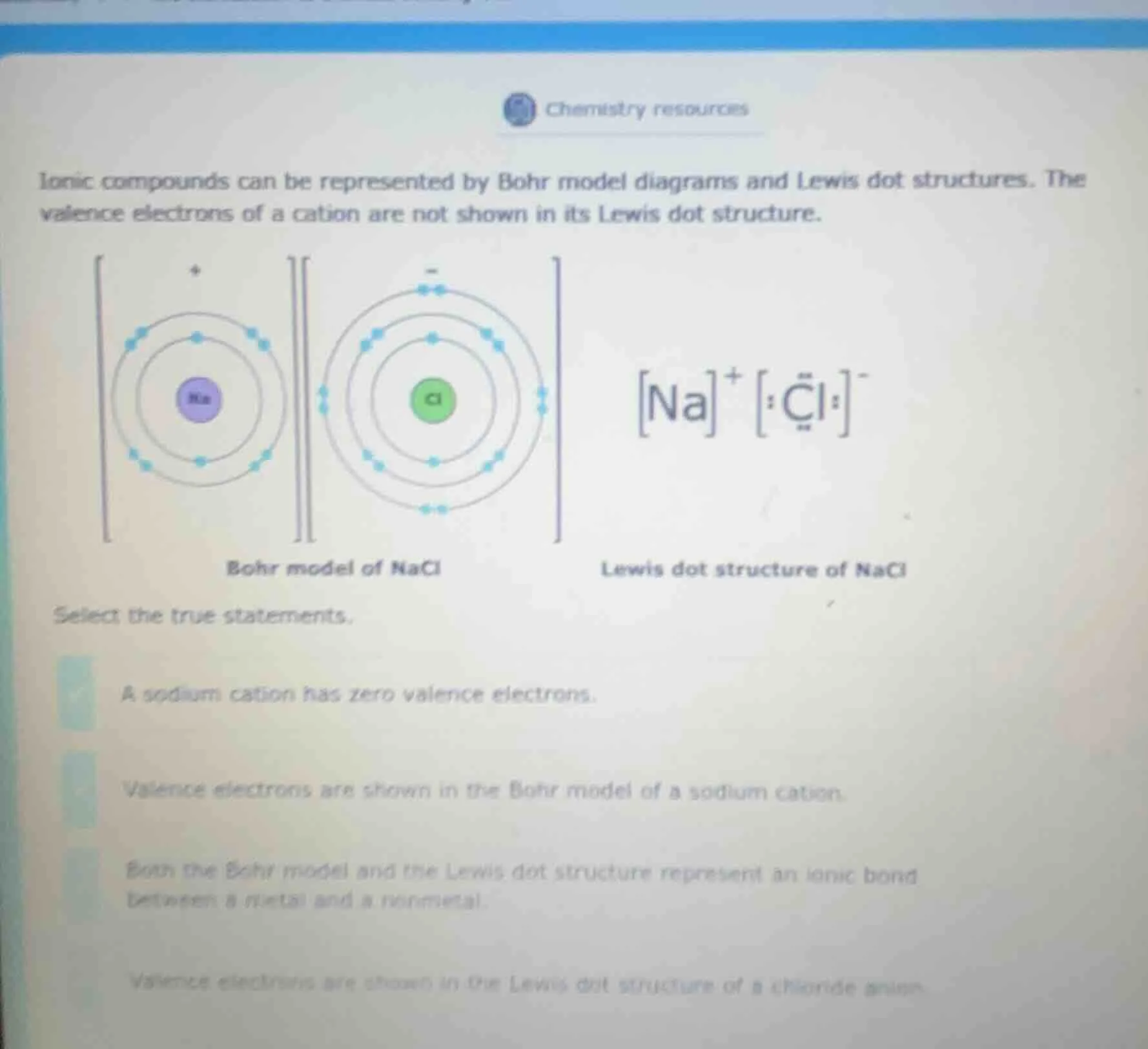
ionic compounds can be represented by bohr model diagrams and lewis dot structures. the valence electrons of a cation are not shown in its lewis dot structure.
bohr model of nacl
lewis dot structure of nacl
select the true statements.
a sodium cation has zero valence electrons.
valence electrons are shown in the bohr model of a sodium cation.
both the bohr model and the lewis dot structure represent an ionic bond between a metal and a nonmetal.
valence electrons are shown in the lewis dot structure of a chloride anion.
- For the first statement: Sodium (Na) has an electron configuration of $1s^22s^22p^63s^1$. When it forms a cation ($Na^+$), it loses its single 3s valence electron, leaving it with 0 valence electrons.
- For the second statement: The Bohr model of the sodium cation shows all remaining electrons (the full inner shells), but these are not valence electrons; valence electrons are the outermost shell electrons, which $Na^+$ no longer has, so this is false.
- For the third statement: NaCl is formed from sodium (a metal) and chlorine (a nonmetal), and both models represent the ionic bond resulting from electron transfer, so this is true.
- For the fourth statement: The Lewis dot structure of the chloride anion ($Cl^-$) shows 8 valence electrons (dots around the Cl symbol), which matches the structure provided, so this is true.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. A sodium cation has zero valence electrons.
C. Both the Bohr model and the Lewis dot structure represent an ionic bond between a metal and a nonmetal.
D. Valence electrons are shown in the Lewis dot structure of a chloride anion.