QUESTION IMAGE
Question
chemistry
unit 3 – quiz 1 practice
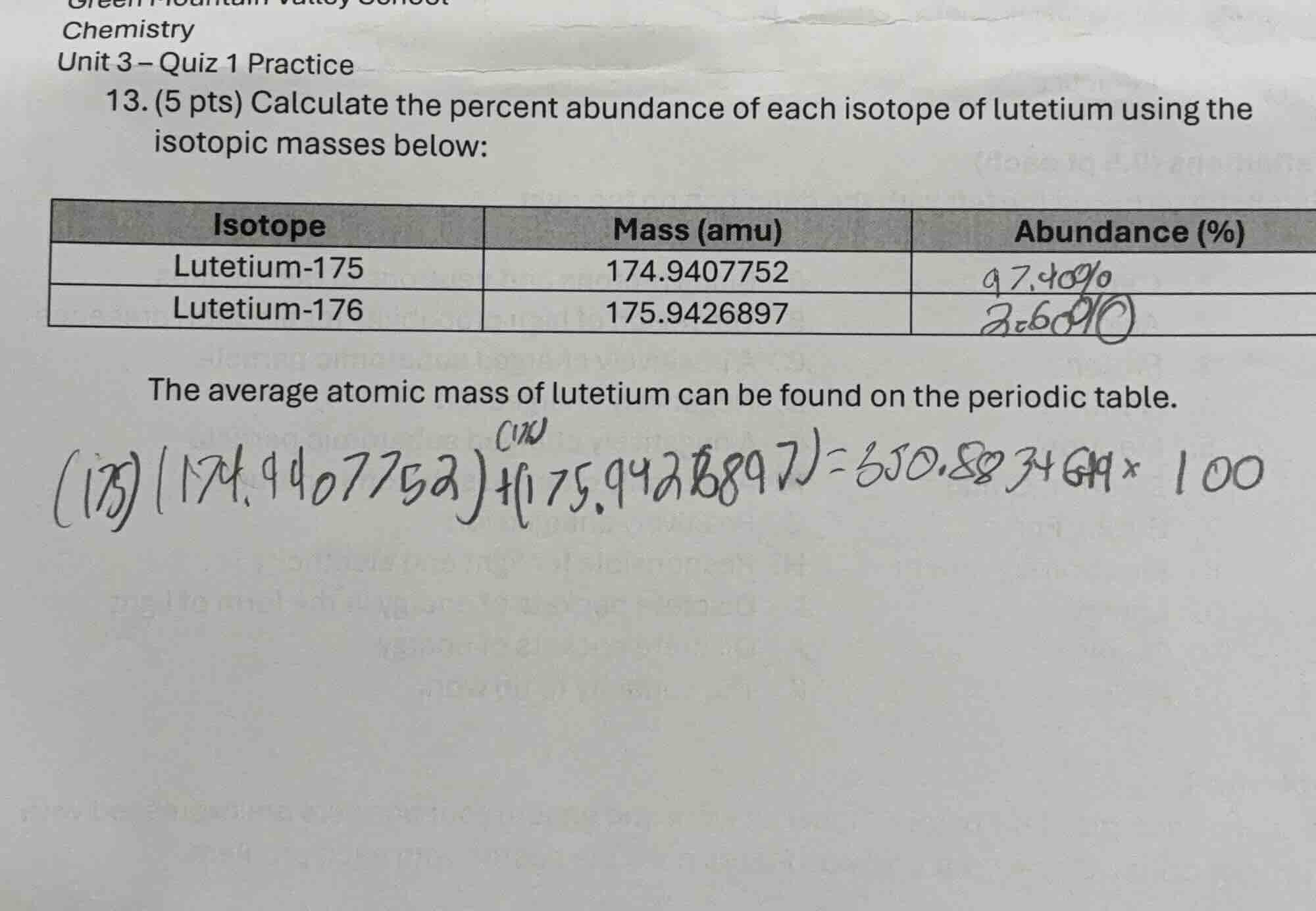
- (5 pts) calculate the percent abundance of each isotope of lutetium using the isotopic masses below:
isotope | mass (amu) | abundance (%)
lutetium-175 | 174.9407752 | 97.40%
lutetium-176 | 175.9426897 | 2.60%
the average atomic mass of lutetium can be found on the periodic table.
$(175)(174.9407752)+(175.9426897)=350.8834649×100$
Step1: Define variables & known values
Let \(x\) = abundance of Lutetium-175 (decimal form), \(1-x\) = abundance of Lutetium-176 (decimal form). Average atomic mass of Lu = \(174.9668\) amu (from periodic table).
Step2: Set up average mass equation
Average atomic mass = (mass of Lu-175 × \(x\)) + (mass of Lu-176 × \((1-x)\))
$$174.9668 = 174.9407752x + 175.9426897(1-x)$$
Step3: Expand and simplify the equation
$$174.9668 = 174.9407752x + 175.9426897 - 175.9426897x$$
$$174.9668 - 175.9426897 = 174.9407752x - 175.9426897x$$
$$-0.9758897 = -1.0019145x$$
Step4: Solve for \(x\)
$$x = \frac{-0.9758897}{-1.0019145} \approx 0.9740$$
Convert to %: \(0.9740 \times 100 = 97.40\%\)
Step5: Find abundance of Lu-176
Abundance of Lu-176 = \(1 - x = 1 - 0.9740 = 0.0260\)
Convert to %: \(0.0260 \times 100 = 2.60\%\)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Percent abundance of Lutetium-175: \(97.40\%\)
Percent abundance of Lutetium-176: \(2.60\%\)