QUESTION IMAGE
Question
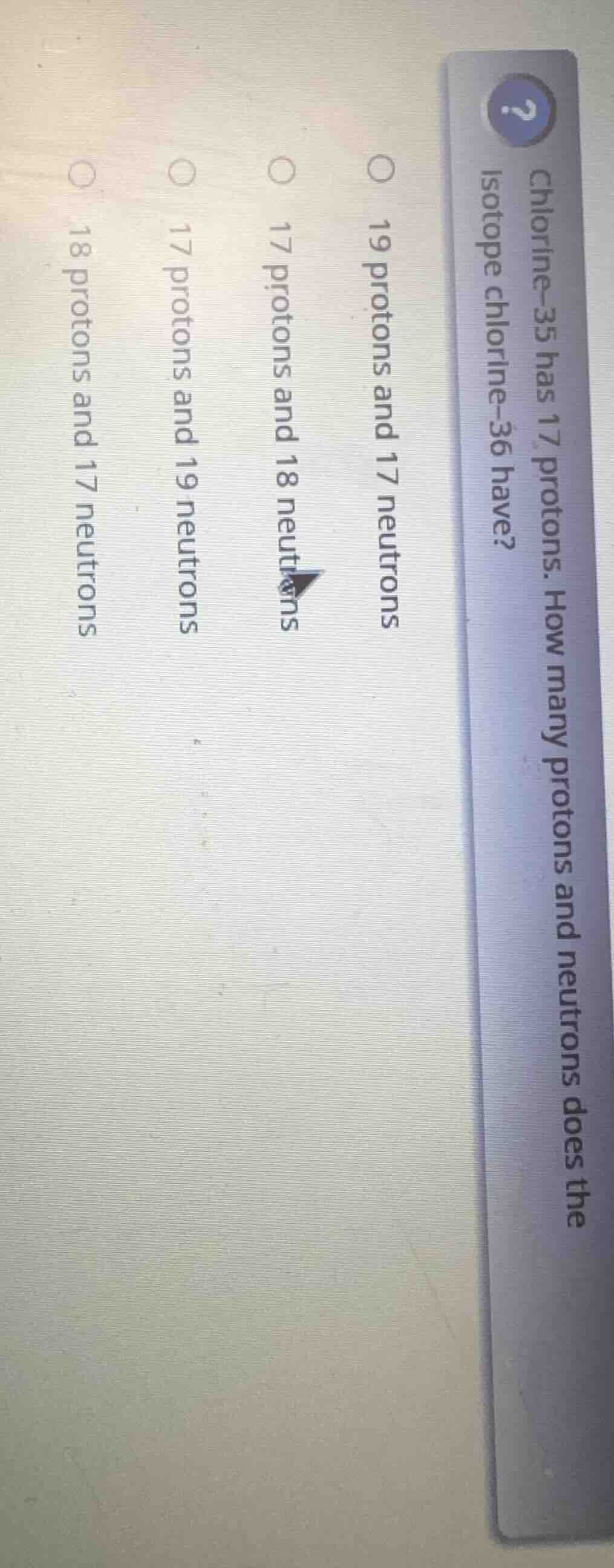
chlorine-35 has 17 protons. how many protons and neutrons does the isotope chlorine-36 have?
19 protons and 17 neutrons
17 protons and 18 neutrons
17 protons and 19 neutrons
18 protons and 17 neutrons
Step1: Confirm proton count
All chlorine isotopes have 17 protons (given for Cl-35, and protons define the element).
Step2: Calculate neutron count
Neutrons = Mass number - Protons. For Cl-36:
$\text{Neutrons} = 36 - 17 = 19$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
17 protons and 19 neutrons