QUESTION IMAGE
Question
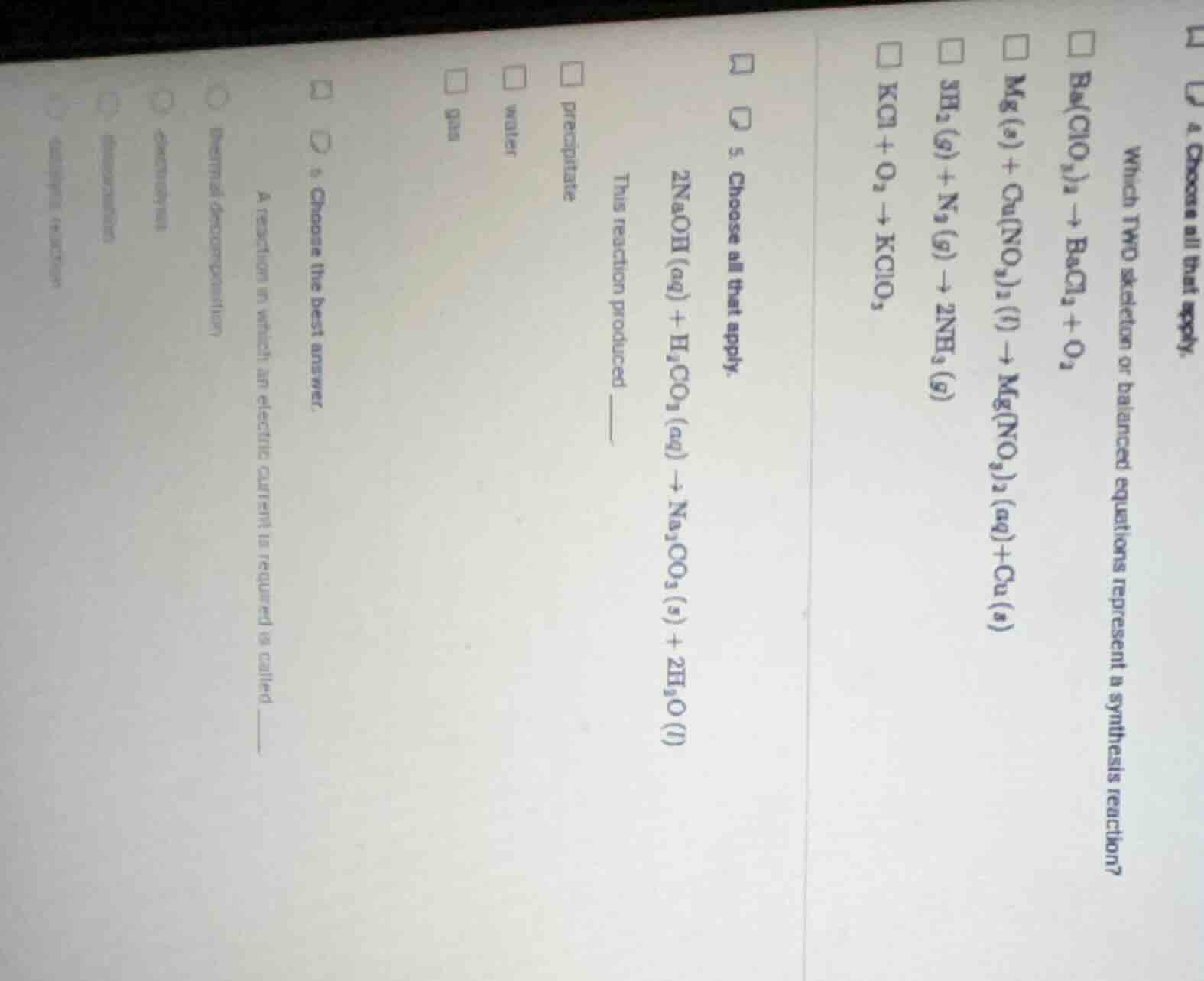
- choose all that apply. which two skeleton or balanced equations represent a synthesis reaction? \\( \square \\ ba(clo_3)_2 \
ightarrow bacl_2 + o_2 \\) \\( \square \\ mg(s) + cu(no_3)_2(l) \
ightarrow mg(no_3)_2(aq) + cu(s) \\) \\( \square \\ 3h_2(g) + n_2(g) \
ightarrow 2nh_3(g) \\) \\( \square \\ kcl + o_3 \
ightarrow kclo_3 \\) 5. choose all that apply. \\( 2naoh(aq) + h_2co_3(aq) \
ightarrow na_2co_3(s) + 2h_2o(l) \\) this reaction produced \\( \square \\) precipitate \\( \square \\) water \\( \square \\) gas 6. choose the best answer. a reaction in which an electric current is required is called \\( \bigcirc \\) thermal decomposition \\( \bigcirc \\) electrolysis \\( \bigcirc \\) omitted text \\( \bigcirc \\) omitted text
Step1: Identify synthesis reactions
A synthesis (combination) reaction follows the pattern $A + B
ightarrow AB$, where two or more reactants form one product.
- $\text{Ba(ClO}_3\text{)}_2
ightarrow \text{BaCl}_2 + \text{O}_2$: This is a decomposition reaction (one reactant → multiple products), not synthesis.
- $\text{Mg}(s) + \text{Cu(NO}_3\text{)}_2(l)
ightarrow \text{Mg(NO}_3\text{)}_2(aq)+\text{Cu}(s)$: This is a single displacement reaction, not synthesis.
- $3\text{H}_2(g) + \text{N}_2(g)
ightarrow 2\text{NH}_3(g)$: Follows $A + B
ightarrow AB$ (two reactants → one product), so it is synthesis.
- $\text{KCl} + \text{O}_2
ightarrow \text{KClO}_3$: Follows $A + B
ightarrow AB$ (two reactants → one product), so it is synthesis.
Step2: Analyze reaction products
For $2\text{NaOH}(aq) + \text{H}_2\text{CO}_3(aq)
ightarrow \text{Na}_2\text{CO}_3(s) + 2\text{H}_2\text{O}(l)$:
- $\text{Na}_2\text{CO}_3(s)$ is a solid precipitate.
- $\text{H}_2\text{O}(l)$ is water.
No gas is produced.
Step3: Identify reaction type with electric current
A reaction requiring an electric current to drive it is defined as electrolysis. Thermal decomposition uses heat, decomposition is a general breakdown reaction, and a synthesis reaction combines reactants.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 4:
C. $3\text{H}_2(g) + \text{N}_2(g)
ightarrow 2\text{NH}_3(g)$
D. $\text{KCl} + \text{O}_2
ightarrow \text{KClO}_3$
Question 5:
precipitate, water
Question 6:
electrolysis