QUESTION IMAGE
Question
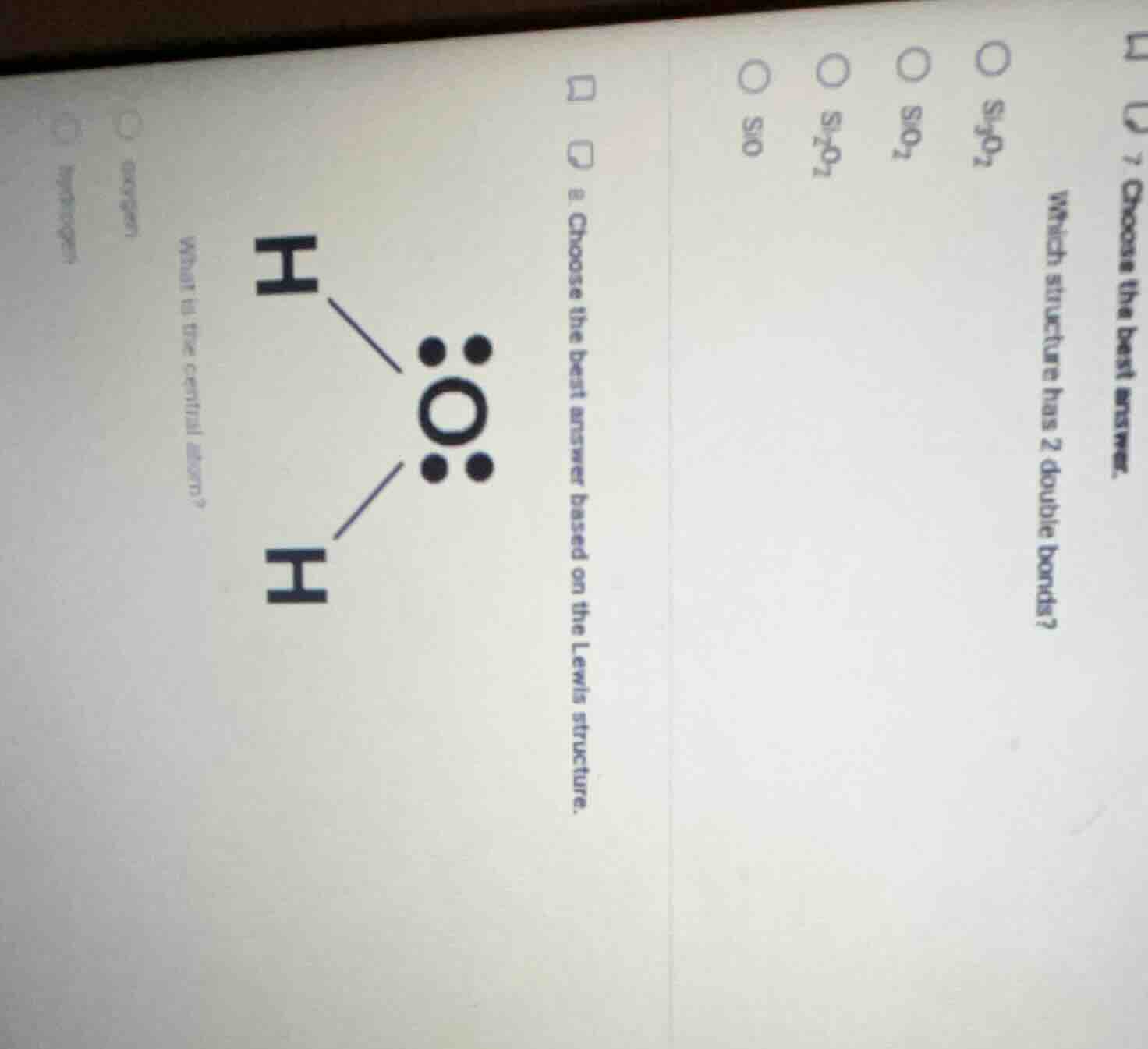
- choose the best answer based on the lewis structure.
what is the central atom?
(options: hydrogen, oxygen)
(another question: 7. choose the best answer.
which structure has 2 double bonds?
options: sio, si₂o₇²⁻, sio₂, si₃o₇²⁻)
Brief Explanations
- For the first question: In the Lewis structure of water ($H_2O$), the central atom is the one bonded to multiple other atoms. Oxygen is bonded to two hydrogen atoms, so it is the central atom.
- For the second question: In $SiO_2$, each silicon atom forms two double covalent bonds with oxygen atoms, resulting in a total of 2 double bonds per silicon center in the molecular structure. The other silicon oxide structures do not have this bonding configuration.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- oxygen
- $SiO_2$