QUESTION IMAGE
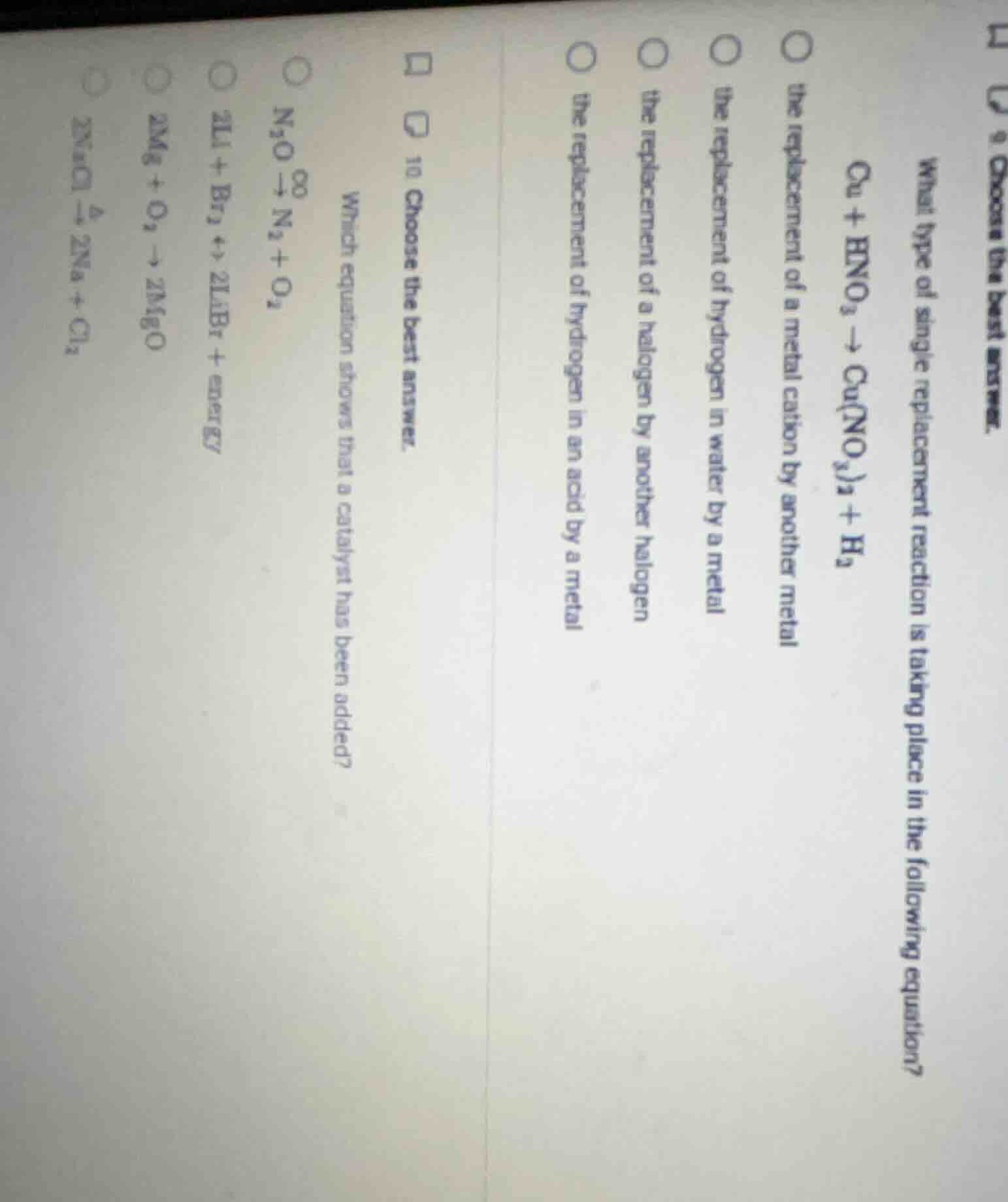
Question
choose the best answer. what type of single replacement reaction is taking place in the following equation? cu + hno₃ → cu(no₃)₂ + h₂ the replacement of a metal cation by another metal the replacement of hydrogen in water by a metal the replacement of a halogen by another halogen the replacement of hydrogen in an acid by a metal 10 choose the best answer. which equation shows that a catalyst has been added? n₂o → n₂ + o₂ 2li + br₂ + energy → 2libr 2mg + o₂ → 2mgo 2nacl xlongequal{delta} 2na + cl₂
For Question 9:
In the reaction $\text{Cu} + \text{HNO}_3
ightarrow \text{Cu(NO}_3\text{)}_2 + \text{H}_2$, copper (a metal) replaces hydrogen in nitric acid (an acid), forming a copper nitrate salt and hydrogen gas. This matches the definition of a single replacement where a metal replaces hydrogen in an acid.
A catalyst speeds up a reaction without being consumed, shown above the reaction arrow ($
ightarrow$). In $\text{N}_2\text{O} \xrightarrow{^\text{Pt}} \text{N}_2 + \text{O}_2$, Pt (platinum) is the catalyst, as it is positioned above the arrow indicating it facilitates the reaction but is not a reactant or product. Other equations show energy input, synthesis, or decomposition without a catalyst marker.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. the replacement of hydrogen in an acid by a metal
---