QUESTION IMAGE
Question
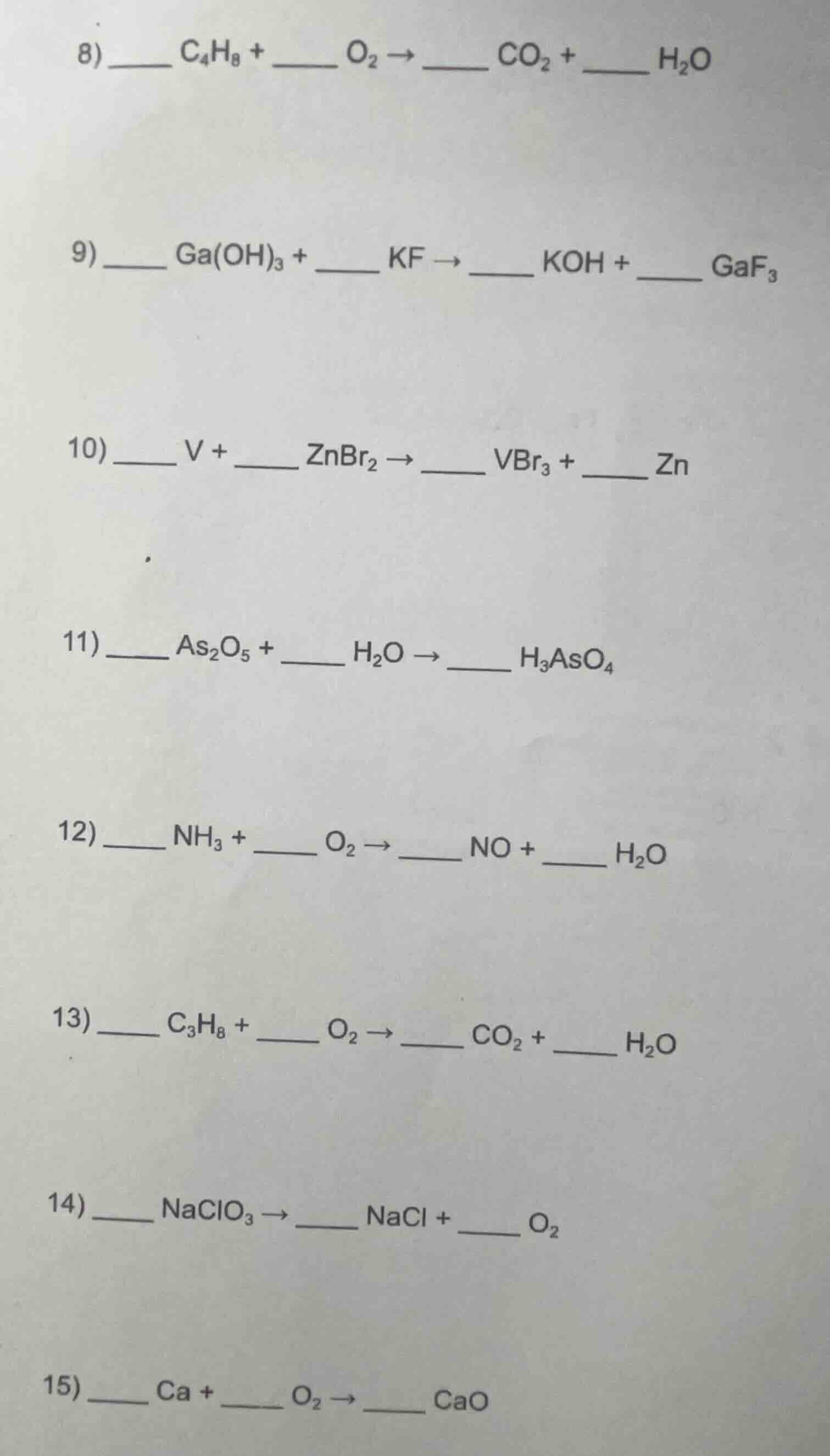
- \underline{\quad\quad} c₄h₈ + \underline{\quad\quad} o₂ → \underline{\quad\quad} co₂ + \underline{\quad\quad} h₂o\
\
- \underline{\quad\quad} ga(oh)₃ + \underline{\quad\quad} kf → \underline{\quad\quad} koh + \underline{\quad\quad} gaf₃\
\
- \underline{\quad\quad} v + \underline{\quad\quad} znbr₂ → \underline{\quad\quad} vbr₃ + \underline{\quad\quad} zn\
\
- \underline{\quad\quad} as₂o₅ + \underline{\quad\quad} h₂o → \underline{\quad\quad} h₃aso₄\
\
- \underline{\quad\quad} nh₃ + \underline{\quad\quad} o₂ → \underline{\quad\quad} no + \underline{\quad\quad} h₂o\
\
- \underline{\quad\quad} c₃h₈ + \underline{\quad\quad} o₂ → \underline{\quad\quad} co₂ + \underline{\quad\quad} h₂o\
\
- \underline{\quad\quad} naclo₃ → \underline{\quad\quad} nacl + \underline{\quad\quad} o₂\
\
- \underline{\quad\quad} ca + \underline{\quad\quad} o₂ → \underline{\quad\quad} cao
Problem 8:
Step1: Balance Carbon atoms
There are 4 C in \( C_4H_8 \), so we put 4 in front of \( CO_2 \).
\( 1\ C_4H_8 + \_\_ O_2
ightarrow 4\ CO_2 + \_\_ H_2O \)
Step2: Balance Hydrogen atoms
There are 8 H in \( C_4H_8 \), so we put 4 in front of \( H_2O \) (since each \( H_2O \) has 2 H).
\( 1\ C_4H_8 + \_\_ O_2
ightarrow 4\ CO_2 + 4\ H_2O \)
Step3: Balance Oxygen atoms
On the right, we have \( 4\times2 + 4\times1 = 12 \) O. So we need 6 \( O_2 \) (since each \( O_2 \) has 2 O).
\( 1\ C_4H_8 + 6\ O_2
ightarrow 4\ CO_2 + 4\ H_2O \)
Step1: Balance Fluorine atoms
There are 3 F in \( GaF_3 \), so we put 3 in front of \( KF \).
\( \_\_ Ga(OH)_3 + 3\ KF
ightarrow \_\_ KOH + 1\ GaF_3 \)
Step2: Balance Potassium atoms
There are 3 K in \( 3\ KF \), so we put 3 in front of \( KOH \).
\( \_\_ Ga(OH)_3 + 3\ KF
ightarrow 3\ KOH + 1\ GaF_3 \)
Step3: Balance Gallium and Oxygen/Hydrogen atoms
There is 1 Ga in \( Ga(OH)_3 \) and 1 in \( GaF_3 \), so we put 1 in front of \( Ga(OH)_3 \). The O and H atoms are already balanced (3 OH in \( Ga(OH)_3 \) and 3 OH in \( 3\ KOH \)).
\( 1\ Ga(OH)_3 + 3\ KF
ightarrow 3\ KOH + 1\ GaF_3 \)
Step1: Balance Bromine atoms
There are 3 Br in \( VBr_3 \) and 2 Br in \( ZnBr_2 \). The least common multiple of 3 and 2 is 6. So we put 2 in front of \( VBr_3 \) and 3 in front of \( ZnBr_2 \).
\( \_\_ V + 3\ ZnBr_2
ightarrow 2\ VBr_3 + \_\_ Zn \)
Step2: Balance Vanadium atoms
There are 2 V in \( 2\ VBr_3 \), so we put 2 in front of \( V \).
\( 2\ V + 3\ ZnBr_2
ightarrow 2\ VBr_3 + \_\_ Zn \)
Step3: Balance Zinc atoms
There are 3 Zn in \( 3\ ZnBr_2 \), so we put 3 in front of \( Zn \).
\( 2\ V + 3\ ZnBr_2
ightarrow 2\ VBr_3 + 3\ Zn \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 1\ C_4H_8 + 6\ O_2
ightarrow 4\ CO_2 + 4\ H_2O \)