QUESTION IMAGE
Question
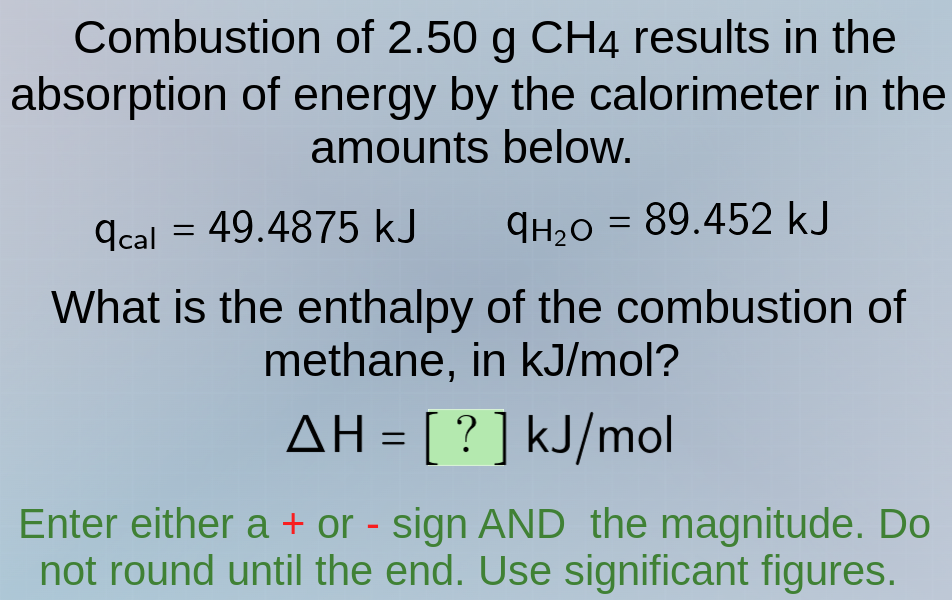
combustion of 2.50 g ch₄ results in the absorption of energy by the calorimeter in the amounts below.
q_cal = 49.4875 kj q_h₂o = 89.452 kj
what is the enthalpy of the combustion of methane, in kj/mol?
δh = ? kj/mol
enter either a + or - sign and the magnitude. do not round until the end. use significant figures.
Step1: Calculate total heat absorbed
The total heat absorbed by the calorimeter and water is \( q_{total}=q_{cal}+q_{H_2O} \).
\( q_{total}=49.4875\ kJ + 89.452\ kJ=138.9395\ kJ \)
Step2: Determine moles of \( CH_4 \)
Molar mass of \( CH_4 \) is \( 12.01 + 4\times1.008 = 16.042\ g/mol \).
Moles of \( CH_4 \), \( n=\frac{mass}{molar\ mass}=\frac{2.50\ g}{16.042\ g/mol}\approx0.15584\ mol \)
Step3: Find enthalpy of combustion
Combustion releases heat, so \( \Delta H \) is negative (since system loses heat).
\( \Delta H=-\frac{q_{total}}{n}=-\frac{138.9395\ kJ}{0.15584\ mol}\approx - 891.5\ kJ/mol \) (considering significant figures, 2.50 has 3 sig figs, so result should have 3 sig figs)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(-892\) (or more precisely \(-891.5\) if we keep more decimals, but with 3 sig figs it's \(-892\))