QUESTION IMAGE
Question
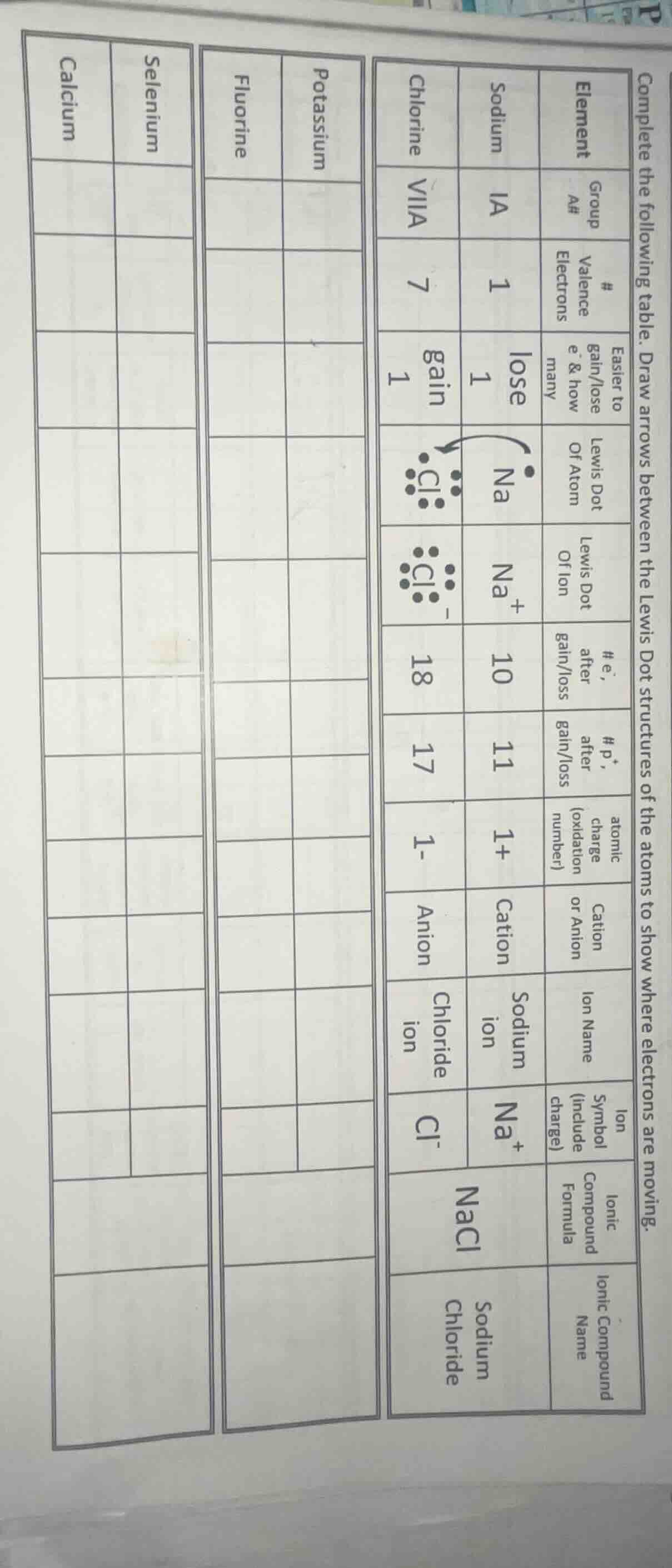
complete the following table. draw arrows between the lewis dot structures of the atoms to show where electrons are moving.
element | sodium | chlorine
group # (a#) | ia | viia
valence electrons | 1 | 7
easier to gain/lose e⁻ & how many | lose 1 | gain 1
lewis dot of atom | na· | :cl:
lewis dot of ion | na⁺ | :cl:⁻
e⁻ after gain/loss | 10 | 18
p⁺ after gain/loss | 11 | 17
atomic charge (oxidation number) | 1+ | 1-
cation or anion | cation | anion
ion name | sodium ion | chloride ion
ion symbol (include charge) | na⁺ | cl⁻
ionic compound formula | nacl |
ionic compound name | sodium chloride |
| potassium | fluorine
|----|----
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| selenium | calcium
|----|----
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
Step1: Fill Potassium (Group IA)
Step1a: Group & valence electrons
Group IA, valence electrons = 1
Step1b: Electron behavior
Lose 1 electron
Step1c: Lewis Dot (Atom/Ion)
Atom: $\dot{K}$; Ion: $K^+$
Step1d: Electrons/protons post-change
$e^-$ after: 18; $p^+$ after: 19
Step1e: Ion properties
Charge: 1+; Cation; Ion Name: Potassium ion; Ion Symbol: $K^+$
Step1f: Ionic compound (with Cl)
Formula: $KCl$; Name: Potassium chloride
Step2: Fill Fluorine (Group VIIA)
Step2a: Group & valence electrons
Group VIIA, valence electrons = 7
Step2b: Electron behavior
Gain 1 electron
Step2c: Lewis Dot (Atom/Ion)
Atom: $\ddot{\underset{\cdot \cdot}{F}} \dot{}$; Ion: $\ddot{\underset{\cdot \cdot}{F}}:^-$
Step2d: Electrons/protons post-change
$e^-$ after: 10; $p^+$ after: 9
Step2e: Ion properties
Charge: 1-; Anion; Ion Name: Fluoride ion; Ion Symbol: $F^-$
Step2f: Ionic compound (with Na)
Formula: $NaF$; Name: Sodium fluoride
Step3: Fill Selenium (Group VIA)
Step3a: Group & valence electrons
Group VIA, valence electrons = 6
Step3b: Electron behavior
Gain 2 electrons
Step3c: Lewis Dot (Atom/Ion)
Atom: $\ddot{\underset{\cdot \cdot}{Se}} \dot{}\dot{}$; Ion: $\ddot{\underset{\cdot \cdot}{Se}}::^{2-}$
Step3d: Electrons/protons post-change
$e^-$ after: 36; $p^+$ after: 34
Step3e: Ion properties
Charge: 2-; Anion; Ion Name: Selenide ion; Ion Symbol: $Se^{2-}$
Step3f: Ionic compound (with Na)
Formula: $Na_2Se$; Name: Sodium selenide
Step4: Fill Calcium (Group IIA)
Step4a: Group & valence electrons
Group IIA, valence electrons = 2
Step4b: Electron behavior
Lose 2 electrons
Step4c: Lewis Dot (Atom/Ion)
Atom: $\dot{Ca}\dot{}$; Ion: $Ca^{2+}$
Step4d: Electrons/protons post-change
$e^-$ after: 18; $p^+$ after: 20
Step4e: Ion properties
Charge: 2+; Cation; Ion Name: Calcium ion; Ion Symbol: $Ca^{2+}$
Step4f: Ionic compound (with Cl)
Formula: $CaCl_2$; Name: Calcium chloride
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Category | Potassium | Fluorine | Selenium | Calcium |
|---|---|---|---|---|
| # Valence Electrons | 1 | 7 | 6 | 2 |
| Easier to gain/lose e⁻ & how many | lose 1 | gain 1 | gain 2 | lose 2 |
| Lewis Dot Of Atom | $\dot{K}$ | $\ddot{\underset{\cdot \cdot}{F}} \dot{}$ | $\ddot{\underset{\cdot \cdot}{Se}} \dot{}\dot{}$ | $\dot{Ca}\dot{}$ |
| Lewis Dot Of Ion | $K^+$ | $\ddot{\underset{\cdot \cdot}{F}}:^-$ | $\ddot{\underset{\cdot \cdot}{Se}}::^{2-}$ | $Ca^{2+}$ |
| # e⁻ after gain/loss | 18 | 10 | 36 | 18 |
| # p⁺ after gain/loss | 19 | 9 | 34 | 20 |
| atomic charge (oxidation number) | 1+ | 1- | 2- | 2+ |
| Cation or Anion | Cation | Anion | Anion | Cation |
| Ion Name | Potassium ion | Fluoride ion | Selenide ion | Calcium ion |
| Ion Symbol (include charge) | $K^+$ | $F^-$ | $Se^{2-}$ | $Ca^{2+}$ |
| Ionic Compound Formula | $KCl$ | $NaF$ | $Na_2Se$ | $CaCl_2$ |
| Ionic Compound Name | Potassium chloride | Sodium fluoride | Sodium selenide | Calcium chloride |
Electron transfer arrows: For K & Cl: arrow from $\dot{K}$ to $\ddot{\underset{\cdot \cdot}{Cl}} \dot{}$; For Na & F: arrow from $\dot{Na}$ to $\ddot{\underset{\cdot \cdot}{F}} \dot{}$; For Na & Se: two arrows from two $\dot{Na}$ to $\ddot{\underset{\cdot \cdot}{Se}} \dot{}\dot{}$; For Ca & Cl: two arrows from $\dot{Ca}\dot{}$ to two $\ddot{\underset{\cdot \cdot}{Cl}} \dot{}$