QUESTION IMAGE
Question
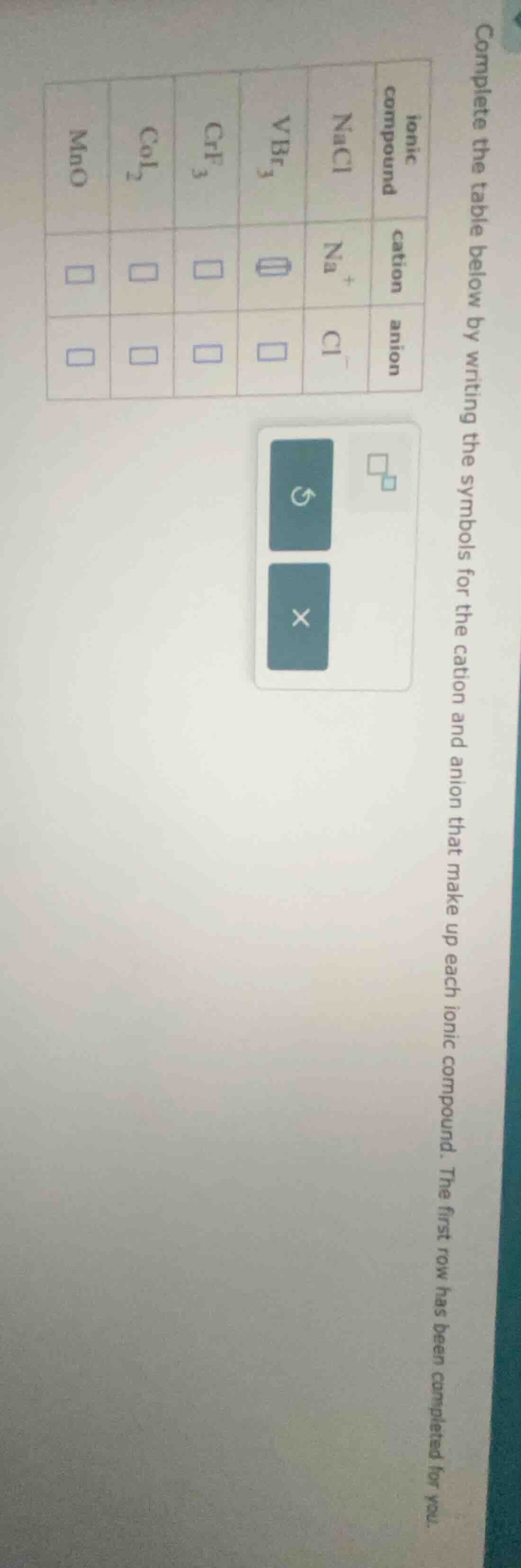
complete the table below by writing the symbols for the cation and anion that make up each ionic compound. the first row has been completed for you.
| ionic compound | cation | anion |
|---|---|---|
| $ce{vbr3}$ | $square$ | $square$ |
| $ce{crf3}$ | $square$ | $square$ |
| $ce{coi2}$ | $square$ | $square$ |
| $ce{mno}$ | $square$ | $square$ |
Step1: Analyze \(VBr_3\)
In ionic compound \(VBr_3\), the cation is \(V^{3+}\) (since the formula has 3 \(Br^-\) ions, and to balance charge, \(V\) has +3 charge) and anion is \(Br^-\).
Step2: Analyze \(CrF_3\)
For \(CrF_3\), the cation is \(Cr^{3+}\) (3 \(F^-\) ions, so \(Cr\) has +3 charge) and anion is \(F^-\).
Step3: Analyze \(CoI_2\)
In \(CoI_2\), the cation is \(Co^{2+}\) (2 \(I^-\) ions, so \(Co\) has +2 charge) and anion is \(I^-\).
Step4: Analyze \(MnO\)
For \(MnO\), the cation is \(Mn^{2+}\) (1 \(O^{2-}\) ion, so \(Mn\) has +2 charge) and anion is \(O^{2-}\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Ionic Compound | Cation | Anion |
|---|---|---|
| \(VBr_3\) | \(V^{3+}\) | \(Br^-\) |
| \(CrF_3\) | \(Cr^{3+}\) | \(F^-\) |
| \(CoI_2\) | \(Co^{2+}\) | \(I^-\) |
| \(MnO\) | \(Mn^{2+}\) | \(O^{2-}\) |