QUESTION IMAGE
Question
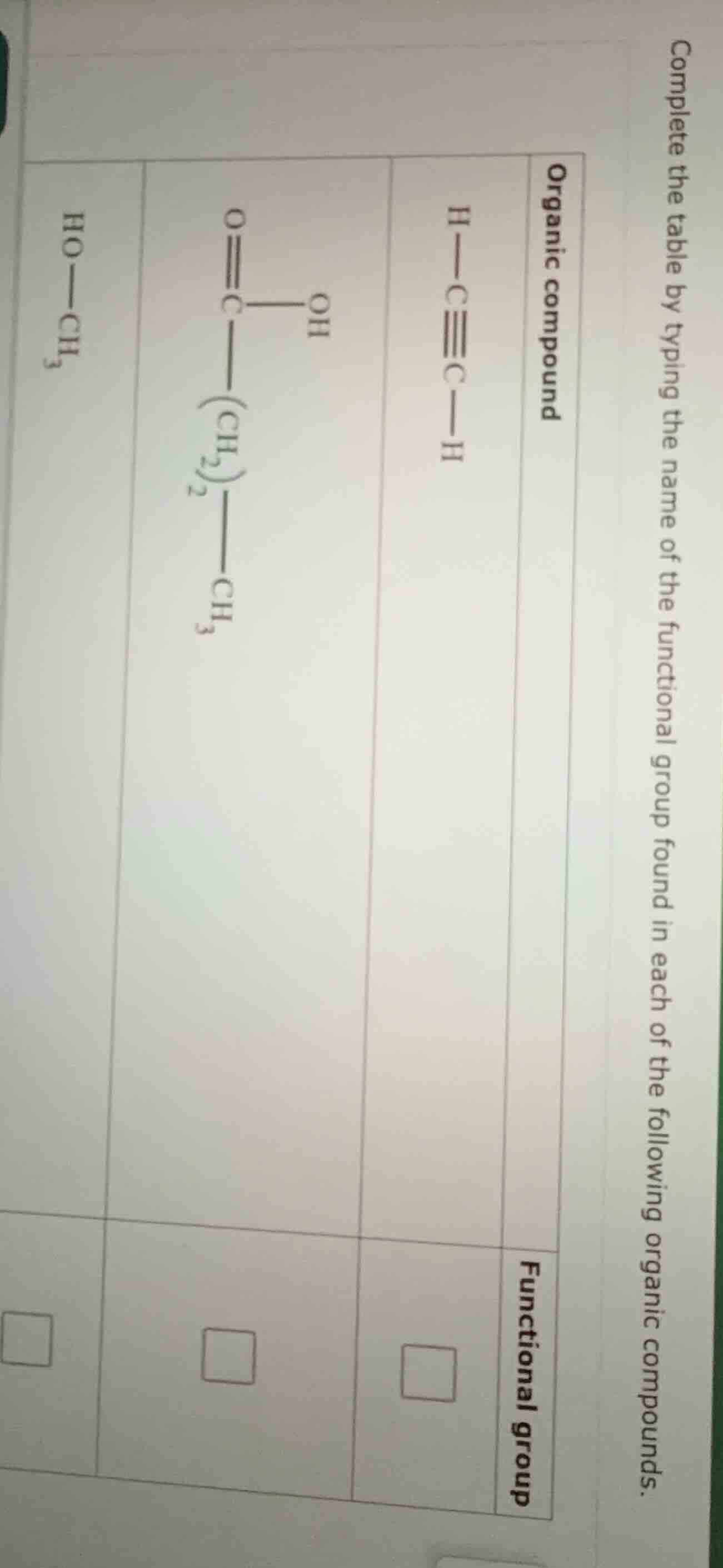
complete the table by typing the name of the functional group found in each of the following organic compounds.
organic compound | functional group
h—c≡c—h |
oh
|o
||
|c—(ch₂)₂—ch₃
||
|o
|
ho—ch₃ |
To solve for the functional groups of each organic compound, we analyze each structure:
1. For $\boldsymbol{\ce{H - C\equiv C - H}}$
- The structure has a carbon - carbon triple bond ($\ce{C\equiv C}$).
- Compounds with a carbon - carbon triple bond are alkynes, and the functional group is an alkyne (or carbon - carbon triple bond).
2. For $\boldsymbol{\ce{OH - C(=O) - (CH_2)_2 - CH_3}}$
- The structure has a $\ce{-COOH}$ (carboxylic acid) group (since it has a carbonyl group $\ce{C=O}$ attached to a hydroxyl group $\ce{-OH}$ and an alkyl chain).
- The functional group here is a carboxylic acid (or carboxyl group, $\ce{-COOH}$).
3. For $\boldsymbol{\ce{HO - CH_3}}$ (methanol - like structure, $\ce{CH_3OH}$)
- The structure has a $\ce{-OH}$ group attached to a carbon atom (the carbon is part of a methyl group here).
- Compounds with a $\ce{-OH}$ group attached to a saturated carbon (not part of a carbonyl group) are alcohols, so the functional group is an alcohol (or hydroxyl group, $\ce{-OH}$).
Filling the Table (assuming a table with rows for each compound and column for functional group):
| Organic compound | Functional group |
|---|---|
| $\ce{OH - C(=O) - (CH_2)_2 - CH_3}$ | Carboxylic Acid (Carboxyl Group) |
| $\ce{HO - CH_3}$ (or $\ce{CH_3OH}$) | Alcohol (Hydroxyl Group) |
If you need the answers in a more concise format (just the names of the functional groups for each compound in order):
- Alkyne (or Carbon - Carbon Triple Bond)
- Carboxylic Acid (or Carboxyl Group)
- Alcohol (or Hydroxyl Group)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve for the functional groups of each organic compound, we analyze each structure:
1. For $\boldsymbol{\ce{H - C\equiv C - H}}$
- The structure has a carbon - carbon triple bond ($\ce{C\equiv C}$).
- Compounds with a carbon - carbon triple bond are alkynes, and the functional group is an alkyne (or carbon - carbon triple bond).
2. For $\boldsymbol{\ce{OH - C(=O) - (CH_2)_2 - CH_3}}$
- The structure has a $\ce{-COOH}$ (carboxylic acid) group (since it has a carbonyl group $\ce{C=O}$ attached to a hydroxyl group $\ce{-OH}$ and an alkyl chain).
- The functional group here is a carboxylic acid (or carboxyl group, $\ce{-COOH}$).
3. For $\boldsymbol{\ce{HO - CH_3}}$ (methanol - like structure, $\ce{CH_3OH}$)
- The structure has a $\ce{-OH}$ group attached to a carbon atom (the carbon is part of a methyl group here).
- Compounds with a $\ce{-OH}$ group attached to a saturated carbon (not part of a carbonyl group) are alcohols, so the functional group is an alcohol (or hydroxyl group, $\ce{-OH}$).
Filling the Table (assuming a table with rows for each compound and column for functional group):
| Organic compound | Functional group |
|---|---|
| $\ce{OH - C(=O) - (CH_2)_2 - CH_3}$ | Carboxylic Acid (Carboxyl Group) |
| $\ce{HO - CH_3}$ (or $\ce{CH_3OH}$) | Alcohol (Hydroxyl Group) |
If you need the answers in a more concise format (just the names of the functional groups for each compound in order):
- Alkyne (or Carbon - Carbon Triple Bond)
- Carboxylic Acid (or Carboxyl Group)
- Alcohol (or Hydroxyl Group)