QUESTION IMAGE
Question
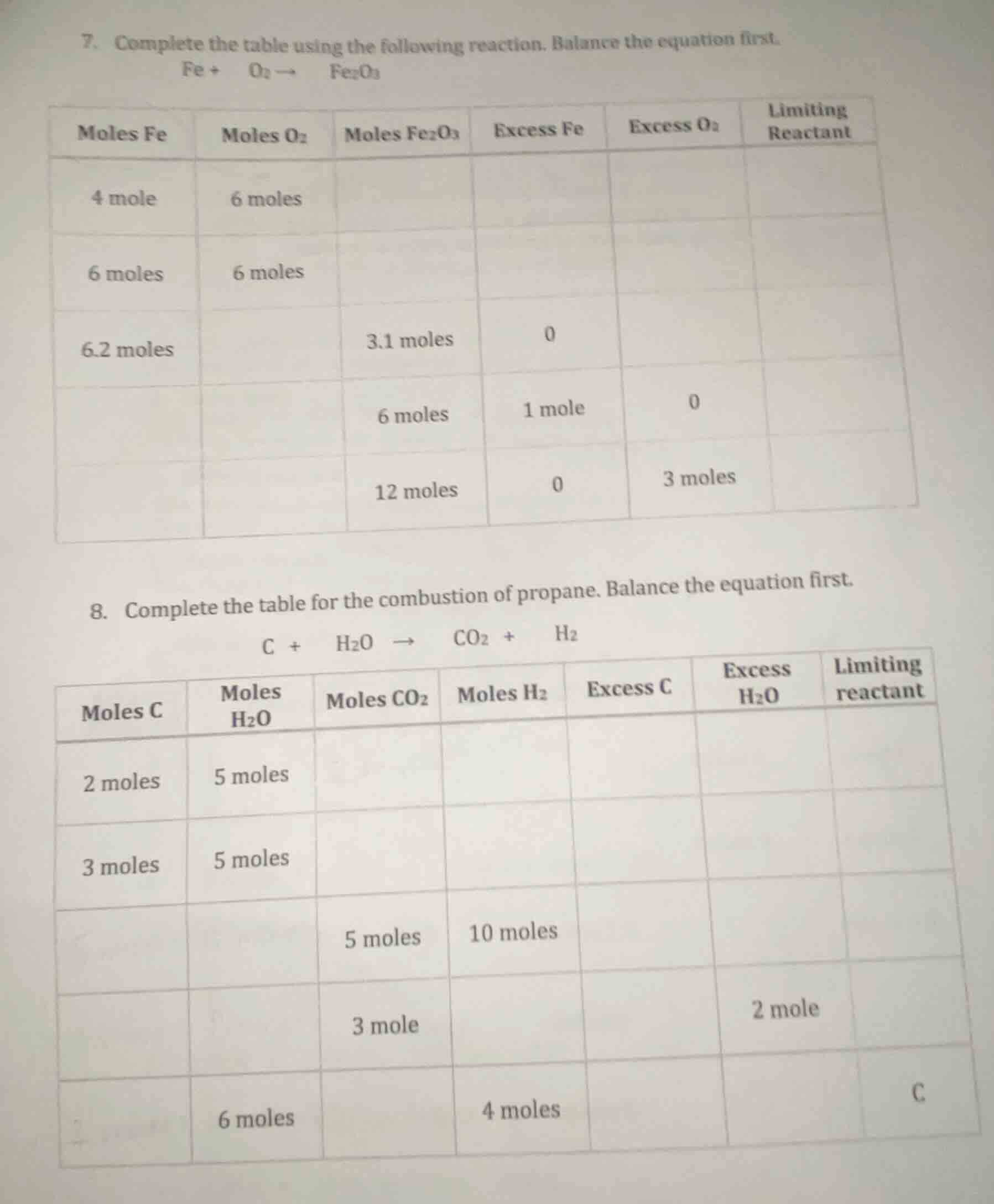
- complete the table using the following reaction. balance the equation first.$ce{fe + o_{2} -> fe_{2}o_{3}}$| moles fe | moles $ce{o_{2}}$ | moles $ce{fe_{2}o_{3}}$ | excess fe | excess $ce{o_{2}}$ | limiting reactant ||----------|----------------|----------------------|-----------|------------------|-------------------|| 4 mole | 6 moles | | | | || 6 moles | 6 moles | | | | || 6.2 moles| | 3.1 moles | 0 | | || | | 6 moles | 1 mole | 0 | || | | 12 moles | 0 | 3 moles | |8. complete the table for the combustion of propane. balance the equation first.$ce{c + h_{2}o -> co_{2} + h_{2}}$| moles c | moles $ce{h_{2}o}$ | moles $ce{co_{2}}$ | moles $ce{h_{2}}$ | excess c | excess $ce{h_{2}o}$ | limiting reactant ||---------|----------------|----------------|----------------|----------|-------------------|-------------------|| 2 moles | 5 moles | | | | | || 3 moles | 5 moles | | | | | || | | 5 moles | 10 moles | | | || | | 3 mole | | | 2 mole | || | 6 moles | | 4 moles | | | c |
Problem 7:
Step1: Balance the reaction
Mole ratio: $\text{Fe}:\text{O}_2:\text{Fe}_2\text{O}_3 = 4:3:2$
---
Row 1: 4 mol Fe, 6 mol O₂
Step2: Find limiting reactant
Fe: $\frac{4}{4}=1$; O₂: $\frac{6}{3}=2$. Fe is limiting.
Step3: Calculate $\text{Fe}_2\text{O}_3$
$\text{Moles of Fe}_2\text{O}_3 = 4 \times \frac{2}{4} = 2$ mol
Step4: Calculate excess O₂
Used O₂: $4 \times \frac{3}{4}=3$ mol; Excess O₂: $6-3=3$ mol
Step5: Identify excess/limiting
Excess Fe = 0; Limiting: Fe
---
Row 2: 6 mol Fe, 6 mol O₂
Step6: Find limiting reactant
Fe: $\frac{6}{4}=1.5$; O₂: $\frac{6}{3}=2$. Fe is limiting.
Step7: Calculate $\text{Fe}_2\text{O}_3$
$\text{Moles of Fe}_2\text{O}_3 = 6 \times \frac{2}{4}=3$ mol
Step8: Calculate excess O₂
Used O₂: $6 \times \frac{3}{4}=4.5$ mol; Excess O₂: $6-4.5=1.5$ mol
Step9: Identify excess/limiting
Excess Fe = 0; Limiting: Fe
---
Row 3: 6.2 mol Fe, 3.1 mol $\text{Fe}_2\text{O}_3$, 0 excess Fe
Step10: Calculate required O₂
$\text{Moles of O}_2 = 3.1 \times \frac{3}{2}=4.65$ mol
Step11: Calculate excess O₂
Excess O₂ = $4.65 - (6.2 \times \frac{3}{4})=0$ (since Fe is fully used, O₂ matches stoichiometry)
Step12: Identify limiting reactant
Limiting: Both (no excess)
---
Row 4: 6 mol $\text{Fe}_2\text{O}_3$, 1 mol excess Fe, 0 excess O₂
Step13: Calculate total Fe
Required Fe: $6 \times \frac{4}{2}=12$ mol; Total Fe: $12+1=13$ mol
Step14: Calculate required O₂
$\text{Moles of O}_2 = 6 \times \frac{3}{2}=9$ mol
Step15: Identify limiting reactant
Limiting: O₂
---
Row 5: 12 mol $\text{Fe}_2\text{O}_3$, 0 excess Fe, 3 mol excess O₂
Step16: Calculate total Fe
$\text{Moles of Fe} = 12 \times \frac{4}{2}=24$ mol
Step17: Calculate total O₂
Used O₂: $12 \times \frac{3}{2}=18$ mol; Total O₂: $18+3=21$ mol
Step18: Identify limiting reactant
Limiting: Fe
---
Step1: Balance the reaction
Mole ratio: $\text{C}:\text{H}_2\text{O}:\text{CO}_2:\text{H}_2 = 1:2:1:2$
---
Row 1: 2 mol C, 5 mol H₂O
Step2: Find limiting reactant
C: $\frac{2}{1}=2$; H₂O: $\frac{5}{2}=2.5$. C is limiting.
Step3: Calculate products
$\text{Moles of CO}_2 = 2 \times \frac{1}{1}=2$ mol; $\text{Moles of H}_2 = 2 \times \frac{2}{1}=4$ mol
Step4: Calculate excess H₂O
Used H₂O: $2 \times 2=4$ mol; Excess H₂O: $5-4=1$ mol
Step5: Identify excess/limiting
Excess C = 0; Limiting: C
---
Row 2: 3 mol C, 5 mol H₂O
Step6: Find limiting reactant
C: $\frac{3}{1}=3$; H₂O: $\frac{5}{2}=2.5$. H₂O is limiting.
Step7: Calculate products
$\text{Moles of CO}_2 = 5 \times \frac{1}{2}=2.5$ mol; $\text{Moles of H}_2 = 5 \times \frac{2}{2}=5$ mol
Step8: Calculate excess C
Used C: $5 \times \frac{1}{2}=2.5$ mol; Excess C: $3-2.5=0.5$ mol
Step9: Identify excess/limiting
Excess H₂O = 0; Limiting: H₂O
---
Row 3: 5 mol CO₂, 10 mol H₂
Step10: Calculate required C
$\text{Moles of C} = 5 \times \frac{1}{1}=5$ mol
Step11: Calculate required H₂O
$\text{Moles of H}_2\text{O} = 5 \times \frac{2}{1}=10$ mol
Step12: Identify excess/limiting
Excess C=0; Excess H₂O=0; Limiting: Both
---
Row 4: 3 mol CO₂, 2 mol excess H₂O
Step13: Calculate required H₂O
Used H₂O: $3 \times 2=6$ mol; Total H₂O: $6+2=8$ mol
Step14: Calculate required C
$\text{Moles of C} = 3 \times \frac{1}{1}=3$ mol
Step15: Calculate $\text{H}_2$ moles
$\text{Moles of H}_2 = 3 \times 2=6$ mol
Step16: Identify limiting reactant
Limiting: C
---
Row 5: 6 mol H₂O, 4 mol H₂, Limiting: C
Step17: Calculate used C/H₂O
Used H₂O: $4 \times \frac{2}{2}=4$ mol; Used C: $4 \times \frac{1}{2}=2$ mol
Step18: Calculate products/excess
$\text{Moles of CO}_2 = 2 \times 1=2$ mol; Excess H₂O: $6-4=2$ mol; Excess C=0
Step19: Confirm total C
Total C = 2 mol (all used)
---
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
(Problem 7 Table):
| Moles Fe | Moles O₂ | Moles Fe₂O₃ | Excess Fe | Excess O₂ | Limiting Reactant |
|---|---|---|---|---|---|
| 6 moles | 6 moles | 3 moles | 0 | 1.5 moles | Fe |
| 6.2 moles | 4.65 moles | 3.1 moles | 0 | 0 | Both |
| 13 moles | 9 moles | 6 moles | 1 mole | 0 | O₂ |
| 24 moles | 21 moles | 12 moles | 0 | 3 moles | Fe |
---