QUESTION IMAGE
Question
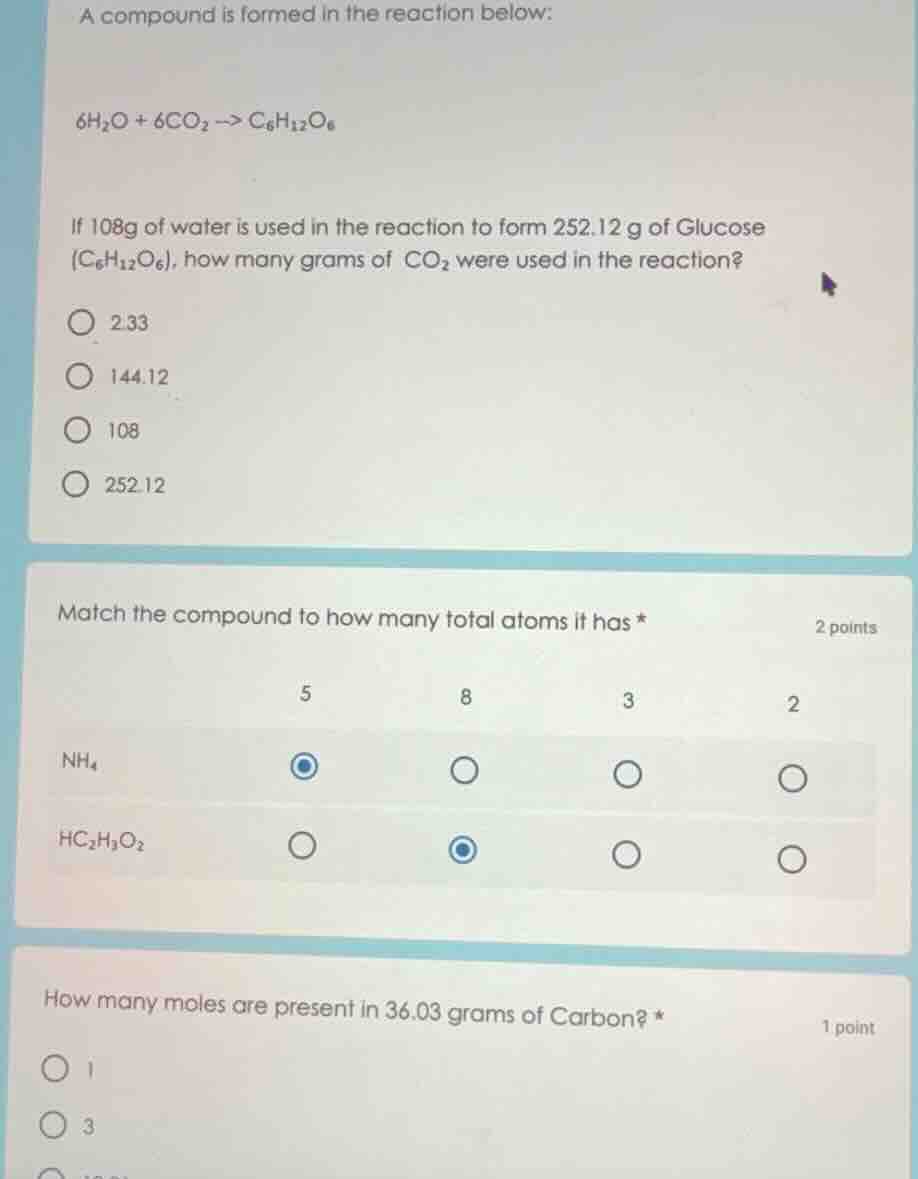
a compound is formed in the reaction below:
$6h_2o + 6co_2 \
ightarrow c_6h_{12}o_6$
if 108g of water is used in the reaction to form 252.12 g of glucose ($c_6h_{12}o_6$), how many grams of $co_2$ were used in the reaction?
\bigcirc 2.33
\bigcirc 144.12
\bigcirc 108
\bigcirc 252.12
match the compound to how many total atoms it has *\t\t2 points
\t\t5\t\t8\t\t3\t\t2
$nh_4$\t\t\bigcirc\t\t\bigcirc\t\t\bigcirc\t\t\bigcirc
$hc_2h_3o_2$\t\t\bigcirc\t\t\bigcirc\t\t\bigcirc\t\t\bigcirc
how many moles are present in 36.03 grams of carbon? *\t\t1 point
\bigcirc 1
\bigcirc 3
\bigcirc
First Sub - Question (Mass of \(CO_2\) used)
Step 1: Recall the law of conservation of mass
In a chemical reaction, the total mass of reactants is equal to the total mass of products. The reaction is \(6H_2O + 6CO_2
ightarrow C_6H_{12}O_6\). Let the mass of \(CO_2\) be \(m_{CO_2}\), mass of \(H_2O\) be \(m_{H_2O}=108\space g\), and mass of \(C_6H_{12}O_6\) be \(m_{C_6H_{12}O_6}=252.12\space g\). So, \(m_{H_2O}+m_{CO_2}=m_{C_6H_{12}O_6}\).
Step 2: Solve for \(m_{CO_2}\)
Rearranging the formula: \(m_{CO_2}=m_{C_6H_{12}O_6}-m_{H_2O}\). Substitute the values: \(m_{CO_2}=252.12 - 108=144.12\space g\).
Second Sub - Question (Matching compounds to total atoms)
- For \(NH_4\) (assuming it's \(NH_4^+\) or a typo for \(NH_4\) - like in a formula), the number of atoms: \(N\) (1) + \(H\) (4) = 5 atoms. So the match with 5 is correct.
- For \(HC_2H_3O_2\) (acetic acid, \(CH_3COOH\) or \(C_2H_4O_2\) - wait, \(HC_2H_3O_2\) has \(H\): 1 + 3 = 4, \(C\): 2, \(O\): 2. Total atoms: 4+2 + 2=8 atoms. So the match with 8 is correct.
Third Sub - Question (Moles of Carbon)
Step 1: Recall the molar mass of Carbon
The molar mass of Carbon (\(C\)) is approximately \(12.01\space g/mol\).
Step 2: Use the formula for moles
Moles (\(n\))=\(\frac{mass}{molar\space mass}\). Given mass = \(36.03\space g\), molar mass of \(C = 12.01\space g/mol\). So, \(n=\frac{36.03}{12.01}=3\space moles\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Mass of \(CO_2\) used: 144.12 g (the option is 144.12)
- For \(NH_4\) - match with 5 is correct; for \(HC_2H_3O_2\) - match with 8 is correct.
- Moles of Carbon: 3 (the option is 3)