QUESTION IMAGE
Question
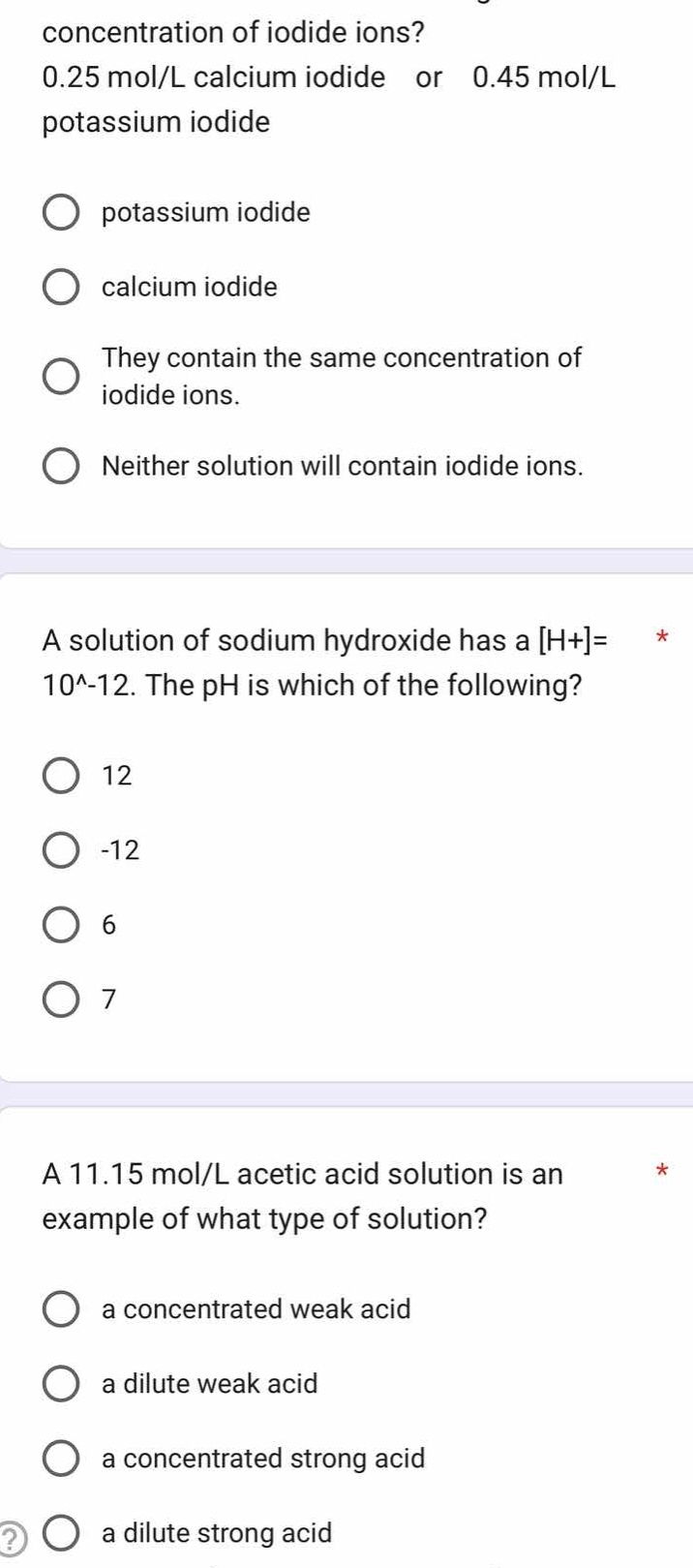
concentration of iodide ions?
0.25 mol/l calcium iodide or 0.45 mol/l
potassium iodide
○ potassium iodide
○ calcium iodide
○ they contain the same concentration of
iodide ions.
○ neither solution will contain iodide ions.
a solution of sodium hydroxide has a h+=
10^-12. the ph is which of the following?
○ 12
○ -12
○ 6
○ 7
a 11.15 mol/l acetic acid solution is an
example of what type of solution?
○ a concentrated weak acid
○ a dilute weak acid
○ a concentrated strong acid
○ a dilute strong acid
Step1: Calculate iodide in calcium iodide
Calcium iodide: $\text{CaI}_2
ightarrow \text{Ca}^{2+} + 2\text{I}^-$, so $[\text{I}^-] = 2 \times 0.25\ \text{mol/L} = 0.50\ \text{mol/L}$
Step2: Calculate iodide in potassium iodide
Potassium iodide: $\text{KI}
ightarrow \text{K}^+ + \text{I}^-$, so $[\text{I}^-] = 1 \times 0.45\ \text{mol/L} = 0.45\ \text{mol/L}$
Step3: Compare iodide concentrations
$0.50\ \text{mol/L} > 0.45\ \text{mol/L}$, so calcium iodide has higher $[\text{I}^-]$
Step4: Calculate pH from $[\text{H}^+]$
$\text{pH} = -\log_{10}[\text{H}^+] = -\log_{10}(10^{-12}) = 12$
Step5: Classify acetic acid solution
Acetic acid is a weak acid (does not fully dissociate); 11.15 mol/L is a high concentration, so it is a concentrated weak acid.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- calcium iodide
- 12
- a concentrated weak acid