QUESTION IMAGE
Question
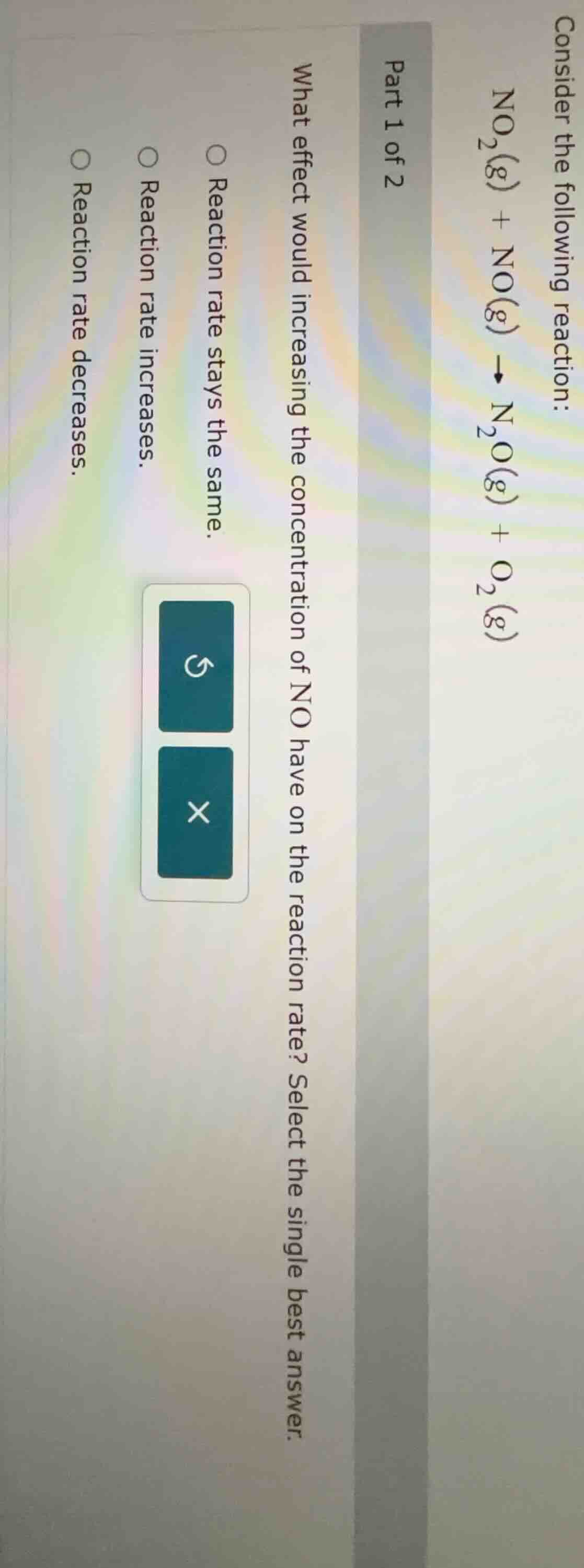
consider the following reaction: no₂(g) + no(g) → n₂o(g) + o₂(g) part 1 of 2 what effect would increasing the concentration of no have on the reaction rate? select the single best answer. reaction rate stays the same. reaction rate increases. reaction rate decreases.
In chemical kinetics, the reaction rate is related to the concentration of reactants. For a reaction involving reactants (like \( \text{NO}_2 \) and \( \text{NO} \) here), increasing the concentration of a reactant (NO in this case) increases the frequency of effective collisions between reactant molecules. This leads to an increase in the reaction rate.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Reaction rate increases.