QUESTION IMAGE
Question
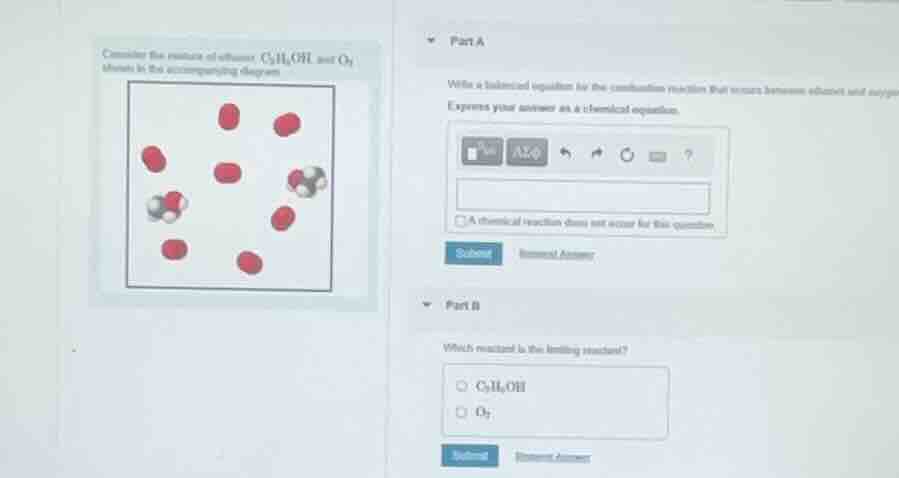
consider the reaction of ethanol $c_2h_5oh$ and $o_2$ shown in the accompanying diagram
part a
write a balanced equation for the combustion reaction that occurs between ethanol and oxygen. express your answer as a chemical equation.
a chemical reaction does not occur for this question.
part b
which reactant is the limiting reactant?
$c_2h_5oh$
$o_2$
Step1: Write unbalanced combustion equation
$\text{C}_2\text{H}_5\text{OH} + \text{O}_2
ightarrow \text{CO}_2 + \text{H}_2\text{O}$
Step2: Balance C atoms first
There are 2 C on left, so add 2 to $\text{CO}_2$:
$\text{C}_2\text{H}_5\text{OH} + \text{O}_2
ightarrow 2\text{CO}_2 + \text{H}_2\text{O}$
Step3: Balance H atoms
There are 6 H on left, so add 3 to $\text{H}_2\text{O}$:
$\text{C}_2\text{H}_5\text{OH} + \text{O}_2
ightarrow 2\text{CO}_2 + 3\text{H}_2\text{O}$
Step4: Balance O atoms
Right side has $2\times2 + 3\times1 = 7$ O. Left side has 1 O in $\text{C}_2\text{H}_5\text{OH}$, so $\text{O}_2$ needs to provide 6 O: add 3 to $\text{O}_2$:
$\text{C}_2\text{H}_5\text{OH} + 3\text{O}_2
ightarrow 2\text{CO}_2 + 3\text{H}_2\text{O}$
Step5: Identify limiting reactant
From the diagram: 2 $\text{C}_2\text{H}_5\text{OH}$ molecules, 8 $\text{O}_2$ molecules.
From balanced equation, 1 $\text{C}_2\text{H}_5\text{OH}$ reacts with 3 $\text{O}_2$.
For 2 $\text{C}_2\text{H}_5\text{OH}$, required $\text{O}_2 = 2\times3 = 6$. We have 8, so $\text{C}_2\text{H}_5\text{OH}$ is used up first.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Part A:
$\text{C}_2\text{H}_5\text{OH}(l) + 3\text{O}_2(g)
ightarrow 2\text{CO}_2(g) + 3\text{H}_2\text{O}(l)$
Part B:
$\text{C}_2\text{H}_5\text{OH}$