QUESTION IMAGE
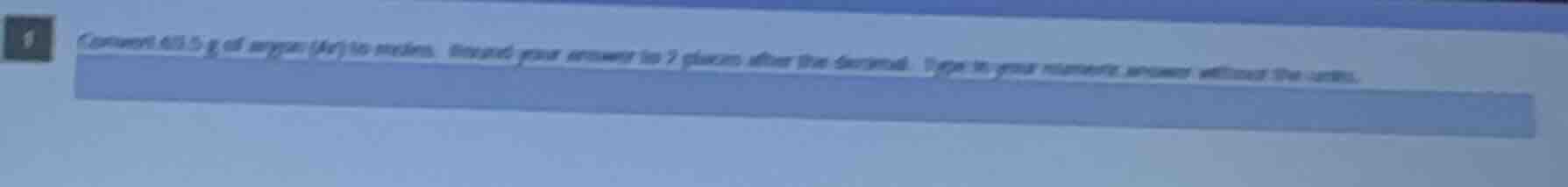
Question
convert 60.5 g of oxygen (o₂) to moles. round your answer to 2 places after the decimal. type in your numeric answer without the units.
Step1: Recall molar mass of Mg
The molar mass of magnesium (Mg) is approximately 24.31 g/mol. This means that one mole of Mg has a mass of 24.31 grams.
Step2: Use the formula for moles
The formula to calculate the number of moles (n) is \( n=\frac{\text{mass (m)}}{\text{molar mass (M)}} \). Here, the mass \( m = 60.5\space g \) and the molar mass \( M=24.31\space g/mol \).
Substitute the values into the formula: \( n=\frac{60.5}{24.31} \)
Calculate the value: \( \frac{60.5}{24.31}\approx2.49 \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2.49