QUESTION IMAGE
Question
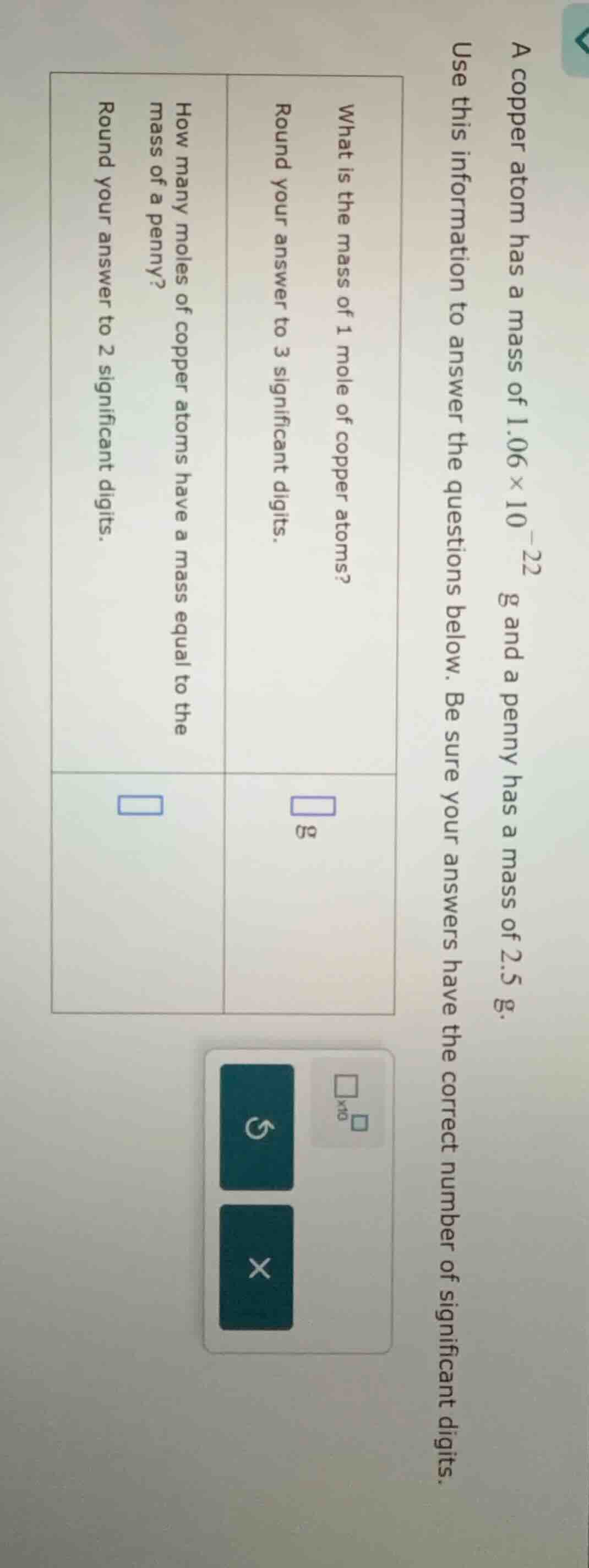
a copper atom has a mass of $1.06 \times 10^{-22}$ g and a penny has a mass of 2.5 g. use this information to answer the questions below. be sure your answers have the correct number of significant digits. \
what is the mass of 1 mole of copper atoms? \
round your answer to 3 significant digits. \
\boxed{} g \
how many moles of copper atoms have a mass equal to the mass of a penny? \
round your answer to 2 significant digits. \
\boxed{}
First Question: Mass of 1 mole of copper atoms
Step1: Recall Avogadro's number
Avogadro's number is \( N_A = 6.022 \times 10^{23} \) atoms/mol, which is the number of atoms in one mole. The mass of one copper atom is \( m = 1.06 \times 10^{-22} \) g. To find the mass of one mole of copper atoms, we multiply the mass of one atom by Avogadro's number.
Step2: Calculate the molar mass
The formula for the mass of one mole (\( M \)) is \( M = m \times N_A \). Substituting the values: \( M=(1.06\times 10^{-22}\ \text{g/atom})\times(6.022\times 10^{23}\ \text{atoms/mol}) \).
First, multiply the coefficients: \( 1.06\times6.022 = 6.38332 \). Then, multiply the powers of 10: \( 10^{-22}\times 10^{23}=10^{1} \). So, \( M = 6.38332\times 10^{1}\ \text{g/mol}=63.8332\ \text{g/mol} \).
Step3: Round to 3 significant digits
Rounding \( 63.8332 \) to 3 significant digits gives \( 63.8 \) g/mol.
Step1: Recall the mass of the penny and molar mass
The mass of the penny is \( m_{\text{penny}} = 2.5 \) g, and the molar mass of copper (from the first part) is \( M = 63.8 \) g/mol. The formula for moles (\( n \)) is \( n=\frac{m_{\text{penny}}}{M} \).
Step2: Calculate the moles
Substitute the values: \( n = \frac{2.5\ \text{g}}{63.8\ \text{g/mol}} \approx 0.0392\ \text{mol} \).
Step3: Round to 2 significant digits
Rounding \( 0.0392 \) to 2 significant digits gives \( 0.039 \) mol (or \( 3.9\times 10^{-2} \) mol).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 63.8 \) g