QUESTION IMAGE
Question
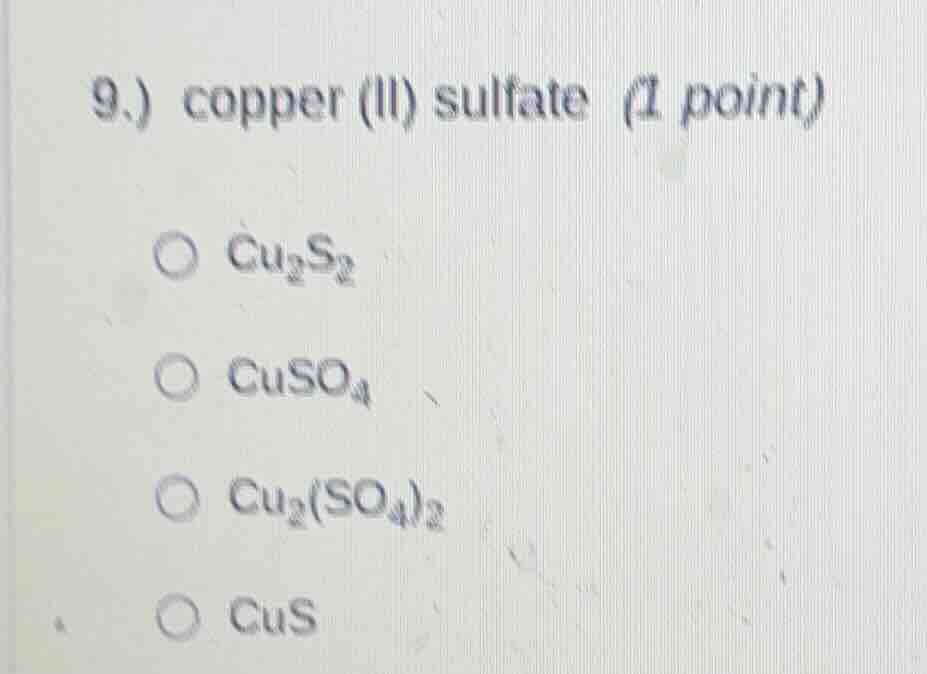
9.) copper (ii) sulfate (1 point)○ $ce{cu_{2}s_{2}}$○ $ce{cuso_{4}}$○ $ce{cu_{2}(so_{4})_{2}}$○ $ce{cus}$
Brief Explanations
Copper (II) has a +2 charge ($\text{Cu}^{2+}$), and the sulfate ion has a -2 charge ($\text{SO}_4^{2-}$). To form a neutral ionic compound, one $\text{Cu}^{2+}$ balances one $\text{SO}_4^{2-}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. $\text{CuSO}_4$