QUESTION IMAGE
Question
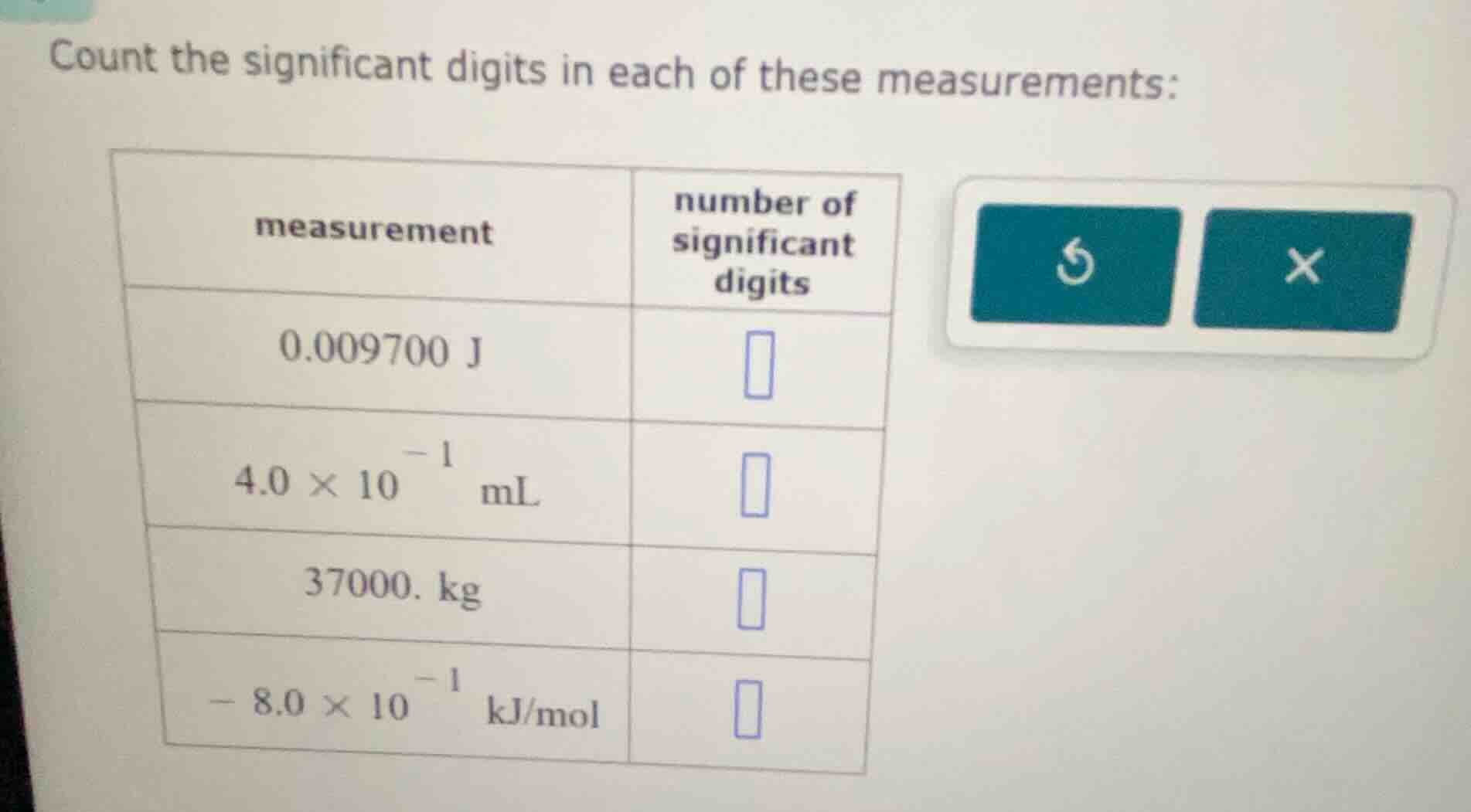
count the significant digits in each of these measurements:
measurement | number of significant digits
0.009700 j |
4.0 × 10⁻¹ ml |
- kg |
− 8.0 × 10⁻¹ kj/mol |
For \( 0.009700 \, \text{J} \):
Step1: Recall significant digit rules
Leading zeros (before the first non - zero digit) are not significant. Non - zero digits are significant. Trailing zeros after a decimal point are significant.
In \( 0.009700 \), the leading zeros (the three zeros before 9) are not significant. The non - zero digits 9 and 7 are significant. The trailing zeros (the two zeros after 7) are also significant.
So we count 9, 7, 0, 0. That's 4 significant digits.
For \( 4.0\times10^{- 1}\, \text{mL} \):
Step1: Recall significant digit rules for scientific notation
In scientific notation \( a\times10^{n} \), the significant digits are in the coefficient \( a \).
Here, the coefficient is 4.0. The 4 is a non - zero digit (significant) and the 0 after the decimal in 4.0 is also significant. So there are 2 significant digits.
For \( 37000. \, \text{kg} \):
Step1: Recall significant digit rules
The decimal point at the end of 37000. indicates that the trailing zeros are significant. The non - zero digits 3 and 7 are significant, and the three zeros after 7 are also significant (because of the decimal point).
So we count 3, 7, 0, 0, 0. That's 5 significant digits.
For \( - 8.0\times10^{-1}\, \text{kJ/mol} \):
Step1: Recall significant digit rules for scientific notation
In scientific notation \( a\times10^{n} \), the significant digits are in the coefficient \( a \). The negative sign does not affect the count of significant digits.
The coefficient is 8.0. The 8 is a non - zero digit (significant) and the 0 after the decimal in 8.0 is also significant. So there are 2 significant digits.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s:
For \( 0.009700 \, \text{J} \): 4
For \( 4.0\times10^{-1}\, \text{mL} \): 2
For \( 37000. \, \text{kg} \): 5
For \( - 8.0\times10^{-1}\, \text{kJ/mol} \): 2