QUESTION IMAGE
Question
covalent bonding
quiz complete
1 2 3 4 5 6 7 8 9 10
time
5
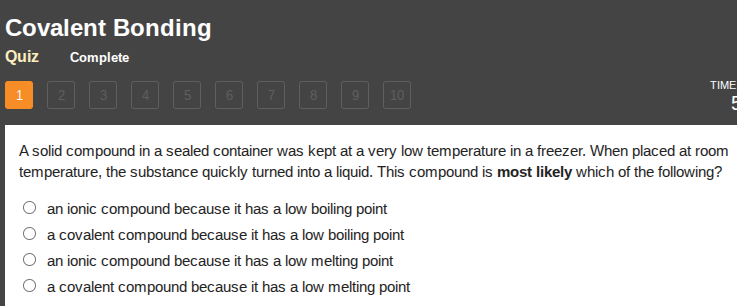
a solid compound in a sealed container was kept at a very low temperature in a freezer. when placed at room temperature, the substance quickly turned into a liquid. this compound is most likely which of the following?
○ an ionic compound because it has a low boiling point
○ a covalent compound because it has a low boiling point
○ an ionic compound because it has a low melting point
○ a covalent compound because it has a low melting point
Ionic compounds typically have high melting points due to strong electrostatic attractions between ions, while covalent compounds (molecular) have weak intermolecular forces, leading to low melting points. The compound melted quickly at room temperature from a low freezer temperature, indicating a low melting point, so it is a covalent compound.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a covalent compound because it has a low melting point