QUESTION IMAGE
Question
covalent bonding
quiz complete
1 2 3 4 5 6 7 8 9 10
time 5
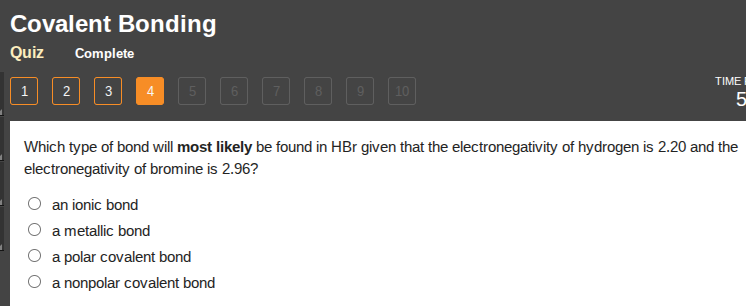
which type of bond will most likely be found in hbr given that the electronegativity of hydrogen is 2.20 and the electronegativity of bromine is 2.96?
an ionic bond
a metallic bond
a polar covalent bond
a nonpolar covalent bond
Brief Explanations
- Calculate electronegativity difference: $2.96 - 2.20 = 0.76$
- Ionic bonds require differences > 1.7, nonpolar covalent < 0.4, and polar covalent falls between 0.4-1.7. This difference fits polar covalent. Metallic bonds occur between metals, which H and Br are not.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a polar covalent bond