QUESTION IMAGE
Question
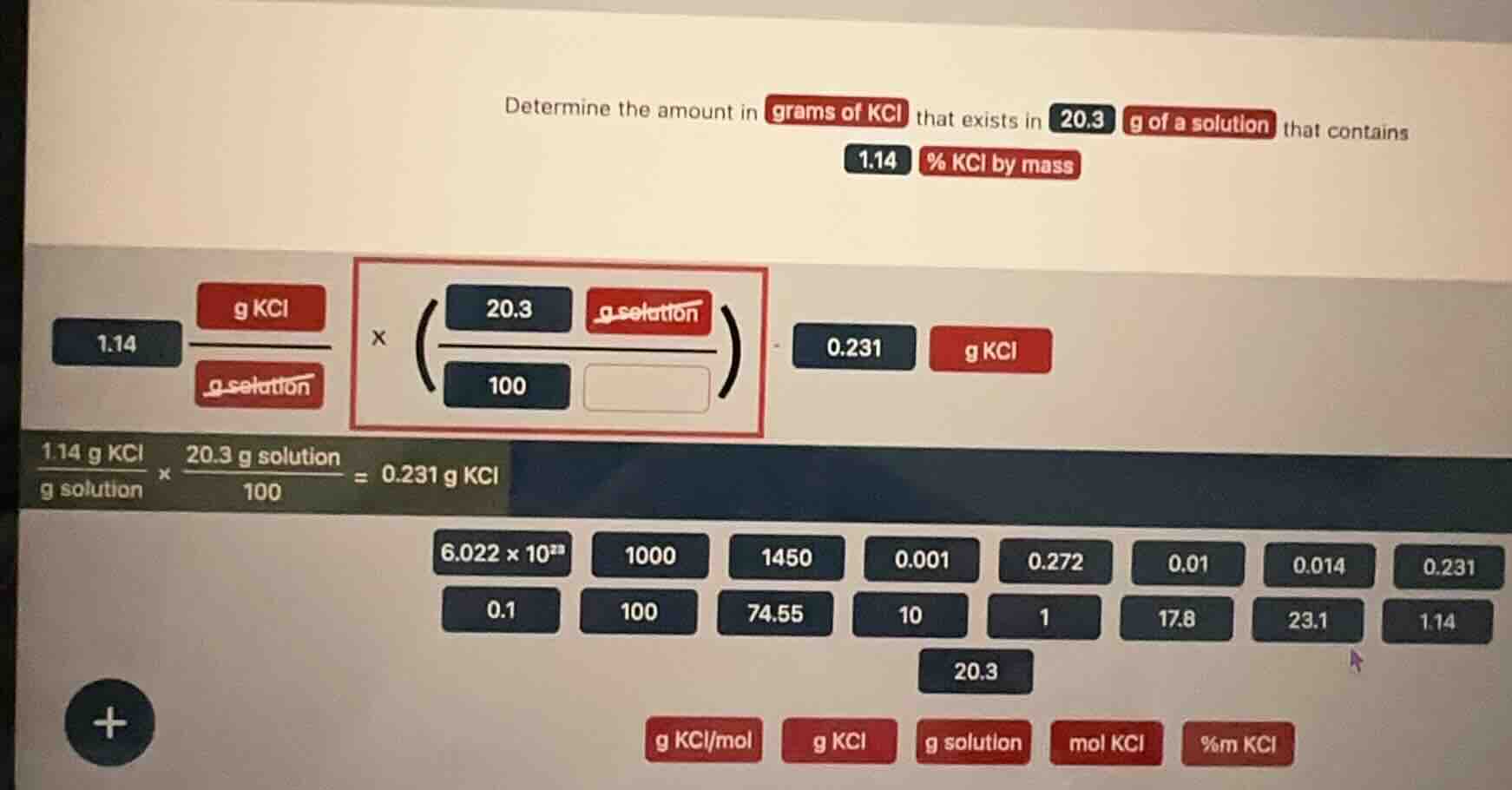
determine the amount in grams of kcl that exists in 20.3 g of a solution that contains 1.14 % kcl by mass
Step1: Set up mass percent formula
The mass percent equation is $\text{mass of KCl} = \frac{\text{mass percent}}{100} \times \text{mass of solution}$
Step2: Substitute given values
Substitute 1.14 for mass percent, 20.3 g for solution mass:
$\text{mass of KCl} = \frac{1.14}{100} \times 20.3$
Step3: Calculate the result
First compute $\frac{1.14}{100}=0.0114$, then multiply by 20.3:
$0.0114 \times 20.3 = 0.23142$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
0.231 g KCl
For the empty box in the red-framed ratio, the correct label is g KCl (to match the unit cancellation logic: $\frac{\text{g KCl}}{\text{g solution}} \times \frac{\text{g solution}}{100}$ leaves g KCl, so the numerator of the second fraction is g solution, denominator 100 pairs with the mass percent, and the missing unit/quantity is g KCl corresponding to the 1.14 value).