QUESTION IMAGE
Question
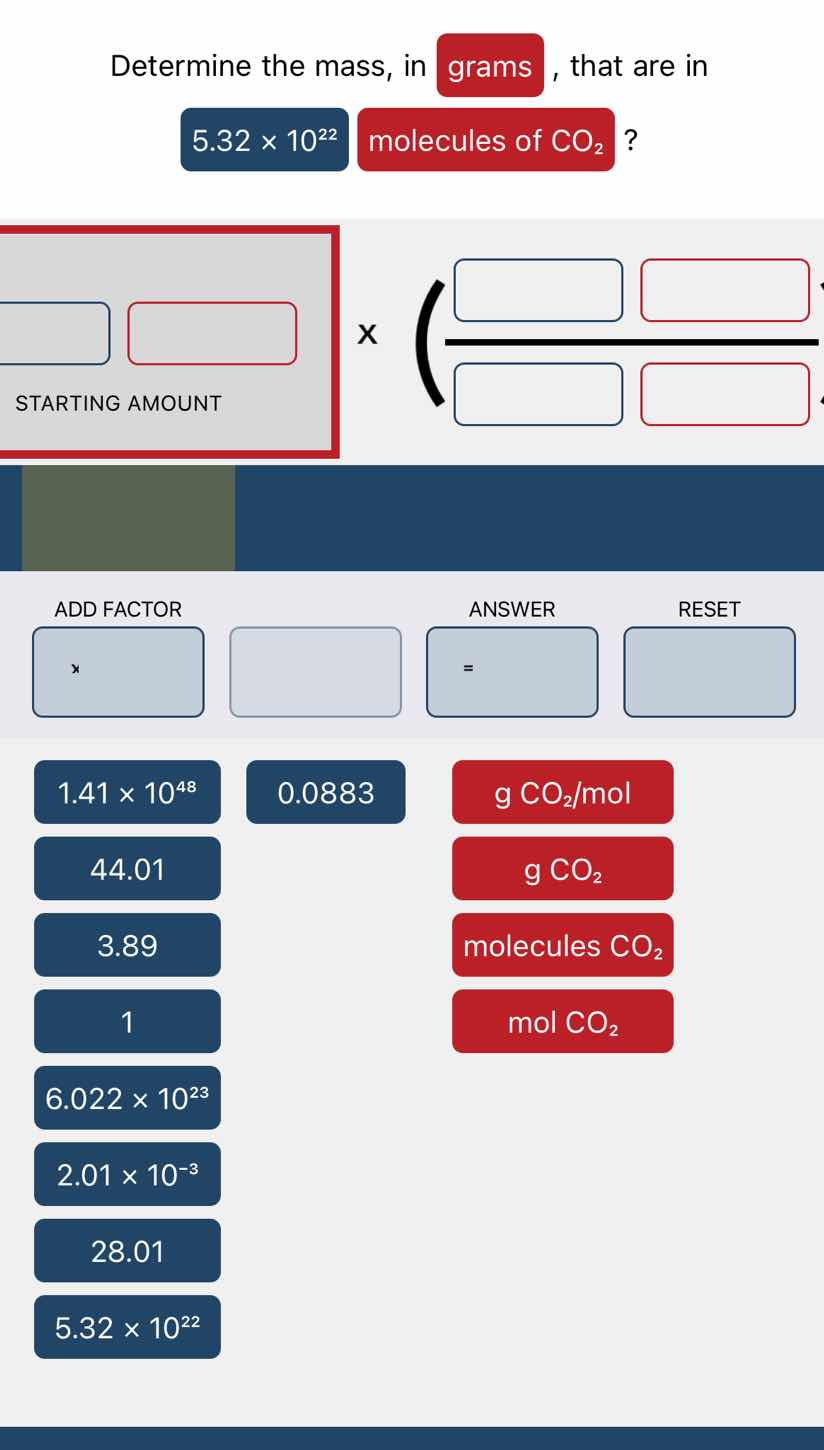
determine the mass, in grams, that are in 5.32 × 10²² molecules of co₂?
starting amount × ( / )
add factor: ×, 1.41 × 10⁴⁸, 0.0883, 44.01, 3.89, 1, 6.022 × 10²³, 2.01 × 10⁻³, 28.01, 5.32 × 10²²
answer: =
reset
units: g co₂/mol, g co₂, molecules co₂, mol co₂
Step1: Set starting amount
$5.32 \times 10^{22} \text{ molecules } \text{CO}_2$
Step2: Convert to moles (Avogadro's number)
$5.32 \times 10^{22} \text{ molecules } \text{CO}_2 \times \frac{1 \text{ mol } \text{CO}_2}{6.022 \times 10^{23} \text{ molecules } \text{CO}_2}$
Step3: Convert moles to grams (molar mass)
$5.32 \times 10^{22} \times \frac{1}{6.022 \times 10^{23}} \times 44.01 \text{ g } \text{CO}_2/\text{mol}$
Step4: Calculate final value
$\frac{5.32 \times 10^{22} \times 44.01}{6.022 \times 10^{23}} = 3.89$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$3.89 \text{ g CO}_2$
(The filled setup is:
Starting amount: $5.32 \times 10^{22}$ molecules $\text{CO}_2$
Conversion factor: $\frac{1 \text{ mol CO}_2}{6.022 \times 10^{23} \text{ molecules CO}_2} \times 44.01 \text{ g CO}_2/\text{mol}$)