QUESTION IMAGE
Question
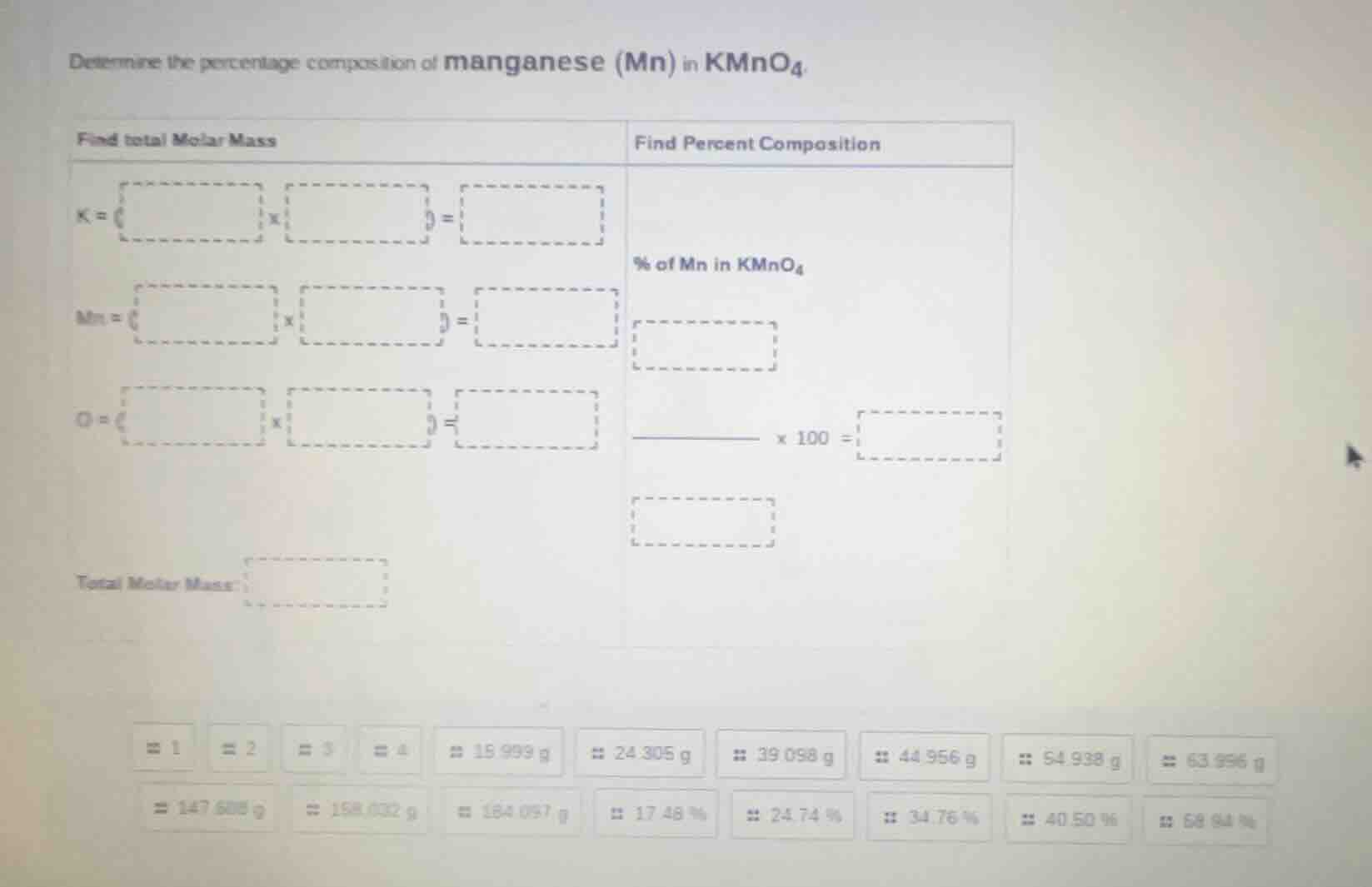
determine the percentage composition of manganese (mn) in kmno₄. find total molar mass find percent composition k = ( ) x ( ) = mn = ( ) x ( ) = o = ( ) x ( ) = total molar mass: % of mn in kmno₄ x 100 =
Step1: Calculate molar mass of K
The atomic mass of K (potassium) is 39.098 g/mol, and there is 1 atom of K in \(KMnO_4\). So, \(K = 39.098\ g/mol\times1 = 39.098\ g/mol\).
Step2: Calculate molar mass of Mn
The atomic mass of Mn (manganese) is 54.938 g/mol, and there is 1 atom of Mn in \(KMnO_4\). So, \(Mn = 54.938\ g/mol\times1 = 54.938\ g/mol\).
Step3: Calculate molar mass of O
The atomic mass of O (oxygen) is 15.999 g/mol, and there are 4 atoms of O in \(KMnO_4\). So, \(O = 15.999\ g/mol\times4 = 63.996\ g/mol\).
Step4: Calculate total molar mass of \(KMnO_4\)
Sum the molar masses of K, Mn, and O: \(Total\ Molar\ Mass = 39.098 + 54.938 + 63.996 = 158.032\ g/mol\).
Step5: Calculate percentage composition of Mn
The formula for percent composition is \(\frac{Mass\ of\ element}{Total\ molar\ mass}\times100\). For Mn, this is \(\frac{54.938}{158.032}\times100\approx34.76\%\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The percentage composition of manganese (Mn) in \(KMnO_4\) is approximately 34.76%.
Filling the boxes:
- For K: \(39.098\) (atomic mass) \(\times\) \(1\) (number of atoms) \(=\) \(39.098\)
- For Mn: \(54.938\) (atomic mass) \(\times\) \(1\) (number of atoms) \(=\) \(54.938\)
- For O: \(15.999\) (atomic mass) \(\times\) \(4\) (number of atoms) \(=\) \(63.996\)
- Total Molar Mass: \(158.032\)
- % of Mn in \(KMnO_4\): \(\frac{54.938}{158.032}\times100 = 34.76\%\)