QUESTION IMAGE
Question
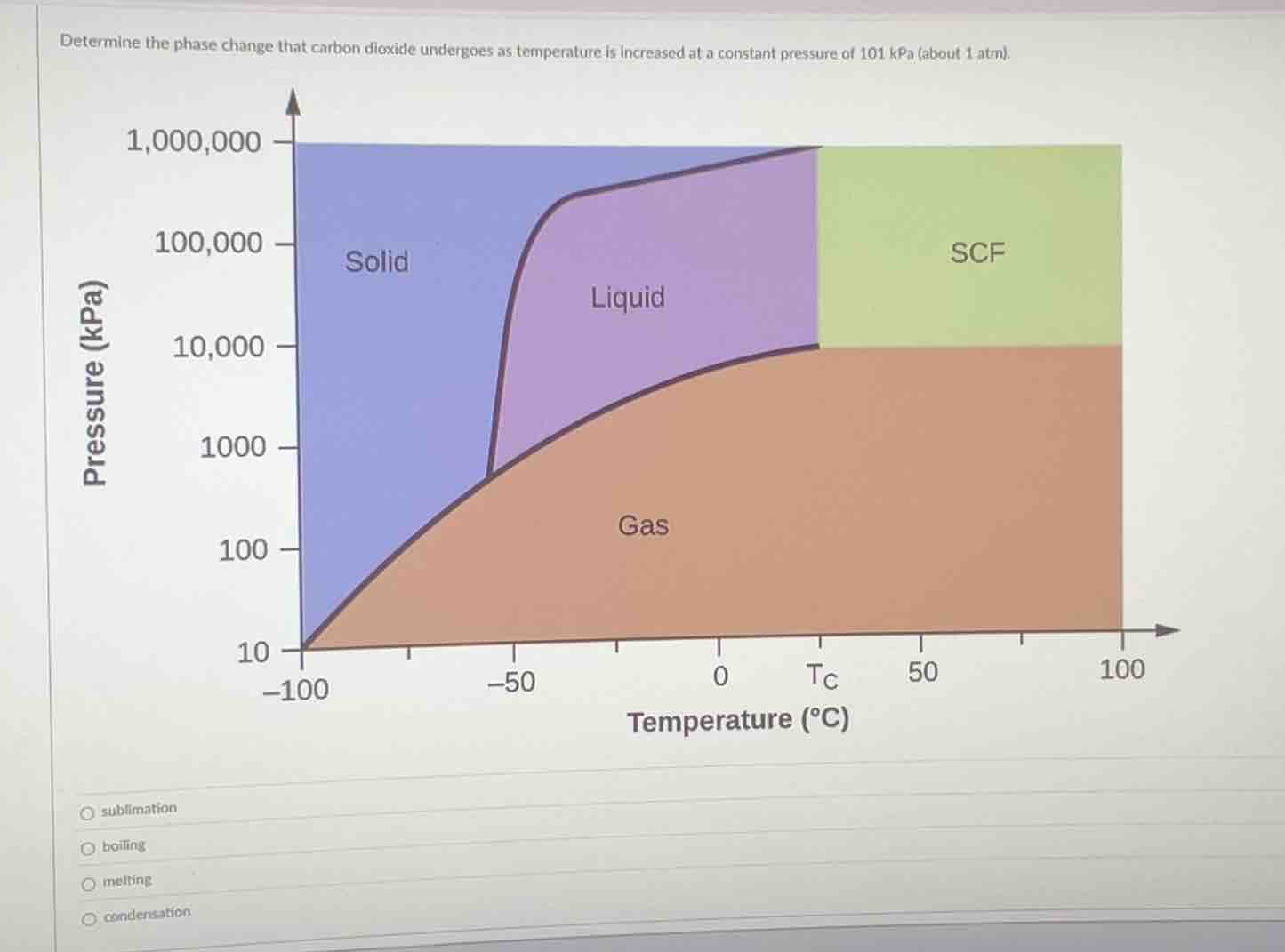
determine the phase change that carbon dioxide undergoes as temperature is increased at a constant pressure of 101 kpa (about 1 atm).
options:
- sublimation
- boiling
- melting
- condensation
Step1: Analyze the phase diagram at 101 kPa
At a constant pressure of 101 kPa (which is in the range of the "Solid" and "Gas" regions' boundary at this pressure), when temperature increases, we observe the phase transition from solid to gas directly.
Step2: Identify the phase change name
The phase change from solid to gas without passing through the liquid phase is called sublimation. Boiling is liquid to gas, melting is solid to liquid, and condensation is gas to liquid, so those don't fit.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
sublimation