QUESTION IMAGE
Question
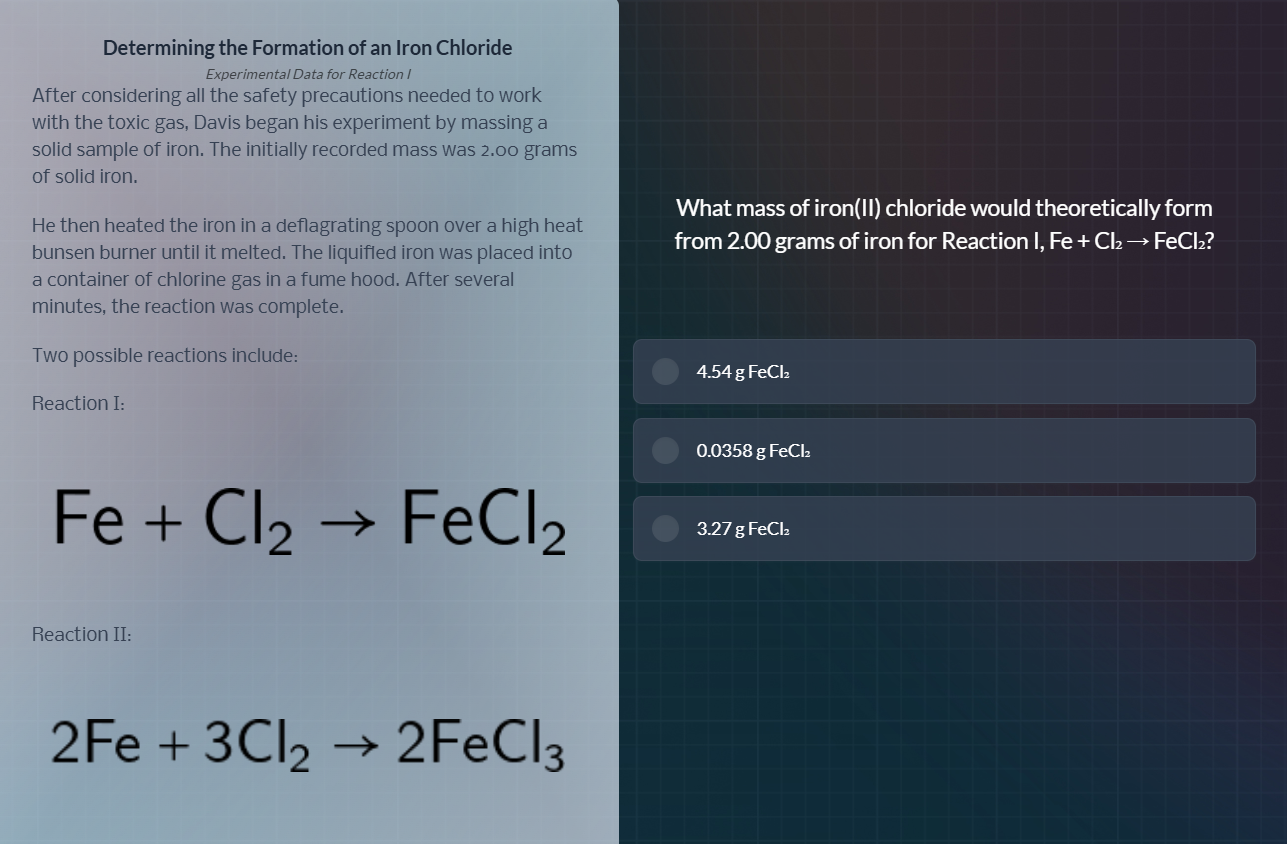
determining the formation of an iron chloride
experimental data for reaction i
after considering all the safety precautions needed to work
with the toxic gas, davis began his experiment by massing a
solid sample of iron. the initially recorded mass was 2.00 grams
of solid iron.
he then heated the iron in a deflagrating spoon over a high heat
bunsen burner until it melted. the liquified iron was placed into
a container of chlorine gas in a fume hood. after several
minutes, the reaction was complete.
two possible reactions include:
reaction i:
fe + cl₂ → fecl₂
reaction ii:
2fe + 3cl₂ → 2fecl₃
what mass of iron(ii) chloride would theoretically form
from 2.00 grams of iron for reaction i, fe + cl₂ → fecl₂?
- 4.54 g fecl₂
- 0.0358 g fecl₂
- 3.27 g fecl₂
Step1: Calculate moles of Fe
Molar mass of Fe is 55.85 g/mol. Moles of Fe = $\frac{mass}{molar\ mass}$ = $\frac{2.00\ g}{55.85\ g/mol}$ ≈ 0.0358 mol.
Step2: Mole ratio from reaction
From $Fe + Cl_2
ightarrow FeCl_2$, mole ratio Fe : $FeCl_2$ is 1:1. So moles of $FeCl_2$ = 0.0358 mol.
Step3: Calculate mass of $FeCl_2$
Molar mass of $FeCl_2$ = 55.85 + 2×35.45 = 126.75 g/mol. Mass = moles × molar mass = 0.0358 mol × 126.75 g/mol ≈ 4.54 g.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
4.54 g FeCl₂