QUESTION IMAGE
Question
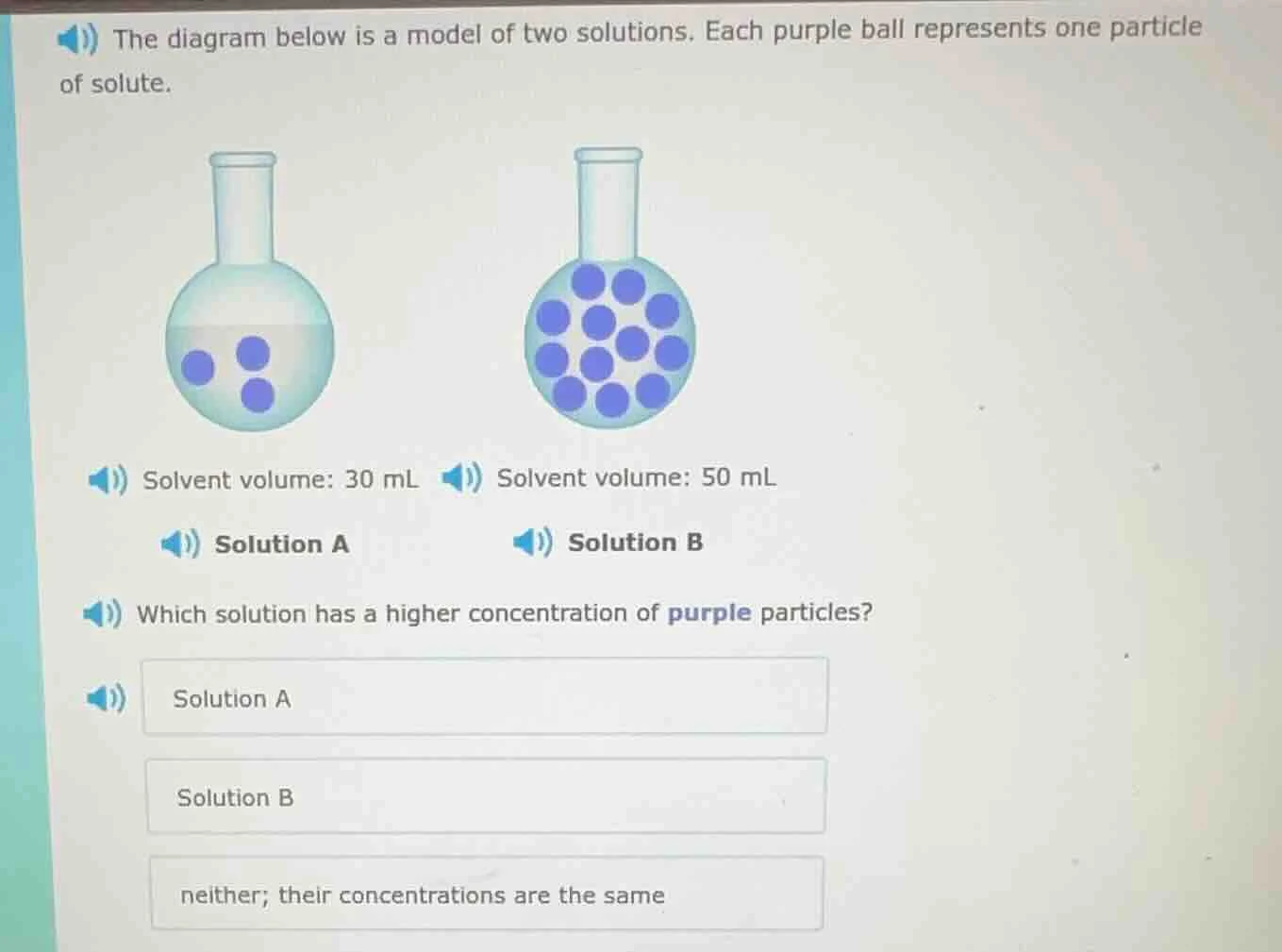
the diagram below is a model of two solutions. each purple ball represents one particle of solute.solvent volume: 30 mlsolution asolvent volume: 50 mlsolution bwhich solution has a higher concentration of purple particles?solution asolution bneither; their concentrations are the same
Step1: Count solute particles for each solution
Solution A: 3 particles; Solution B: 15 particles
Step2: Calculate concentration (particles/mL)
Concentration of A: $\frac{3}{30} = 0.1$ particles/mL
Concentration of B: $\frac{15}{50} = 0.3$ particles/mL
Step3: Compare the two concentrations
$0.3 > 0.1$, so Solution B has higher concentration.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Solution B